Nature doesn’t like symmetry. Our bodies are built of ‘left-handed’ amino acids and ‘right-handed’ sugars and is essentially a giant chiral environment. Everything from our DNA to our taste buds is “handed.” Here the difference between a cure and a toxin is often just a “left-handed” or “right-handed” orientation. Therefore, a drug must be 3D-printed at the molecular level to fit the lock. Asymmetric synthesis is our 3D printer.
What is Asymmetric Synthesis?
Normally, when a reagent attacks a flat (planar) molecule like a ketone, it can hit from the Top or the Bottom with equal probability. In a standard lab reaction, you usually get a Racemic Mixture (50:50). However, since the human body is “chiral” (our receptors and enzymes are like right-handed gloves), we often need only the “right-handed” version of a drug.
In simple terms, Asymmetric Synthesis is the process of turning a non-chiral starting material into a chiral product. It produces one stereoisomer (usually an enantiomer) in more quantity than the other. It is the art of “tilting the playing field” so the reagent is forced to choose one side over the other.
Note: Think of it like a bakery. A normal oven makes round cookies. Asymmetric synthesis is like using a specific mold to make only star-shaped cookies.
Partial Asymmetric Synthesis
In Partial Synthesis, we use a “helper” that is already chiral to guide the reaction. This is like a father holding a child’s hand to help them write—the father’s “handedness” guides the child’s movement. In this type of synthesis, we use a pre-existing chiral center in the molecule to influence the formation of a new one.
A. Chiral Auxiliaries (The Temporary Guide)
We physically attach a chiral molecule (the auxiliary) to our starting material.
- Attachment: The auxiliary (chiral molecule) bonds to the achiral substrate. This chiral buddy acts like a shield
- Reaction: Because the auxiliary is bulky and chiral, it blocks one side of the molecule.
- Removal: Once the new chiral center is formed, we remove the auxiliary and reuse it.
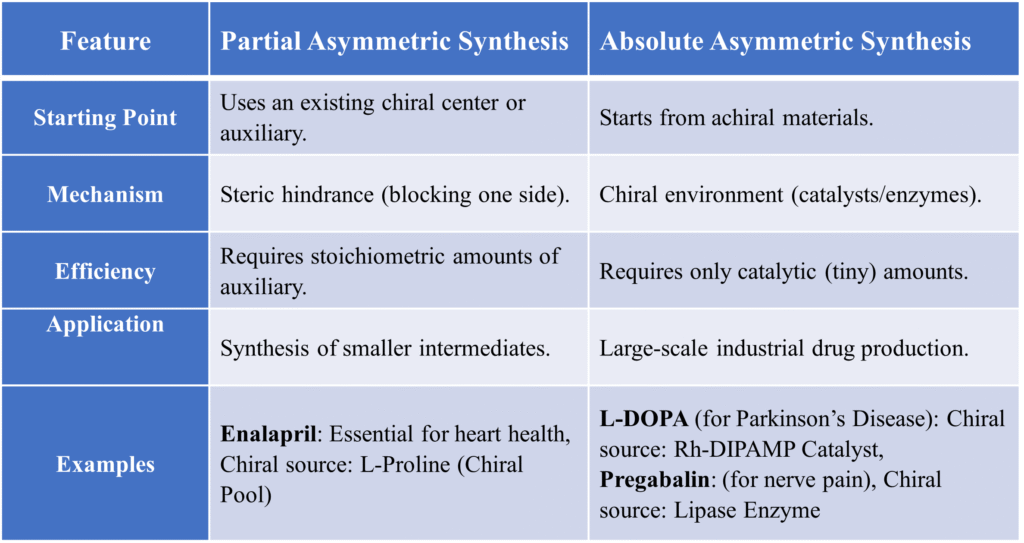
B. Substrate Control: Cram’s Rule
When a molecule already has a chiral center near the reaction site, that center dictates the geometry. It helps us predict which side a reagent will attack based on the size of the groups (Large, Medium, Small) already present on the molecule.
Basic guidelines of Cram’s Rule
The basic steps in Cram’s rule are explained below.
- First of all, assign the Large group(L), Medium(M) and small(S) groups at the chiral center.
- Determine the orientation of Large group with carbonyl group. If L group is syn to carbonyl group then, nucleophile will attack from the side similar to medium group.
- If L group is Anti to carbonyl group then, nucleophile will attack from the side similar to smaller group
Example
Imagine a carbonyl group (C=O) next to a carbon with Large (L), Medium (M), and Small (S) groups. As per the rule, the oxygen of the carbonyl aligns itself between the medium and small groups. The incoming nucleophile will then attack from the side of the small group because there is less “traffic” (steric hindrance).
Absolute Asymmetric Synthesis
Here, we start with achiral materials and use an external source of “asymmetric force” to create a 100% pure enantiomer from scratch.
A. Chiral Catalysts (The “Lego” Approach)
Instead of attaching a helper to every molecule, we use a tiny amount of a chiral catalyst. The catalyst grabs the starting material, holds it in a specific 3D orientation, lets the reaction happen, and then lets go to grab the next one.
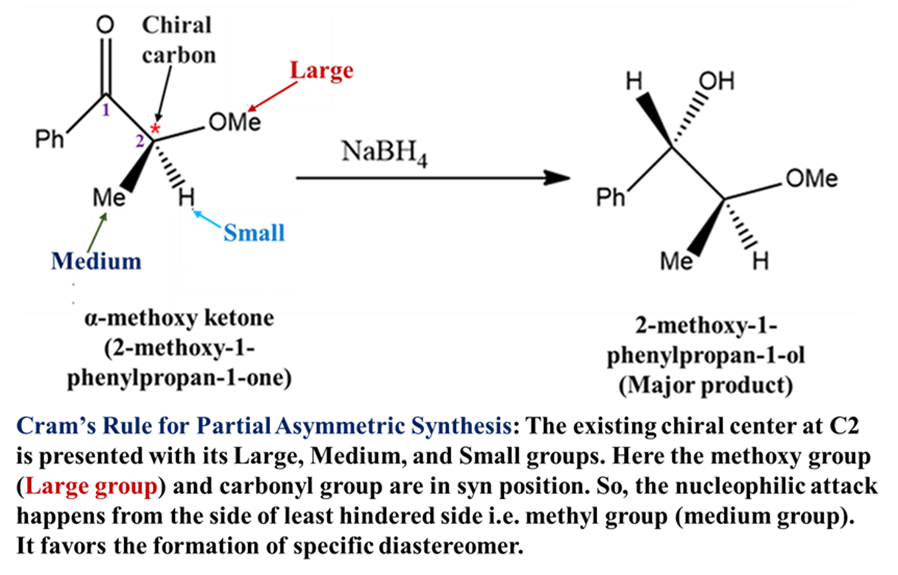
Example: The synthesis of L-DOPA (for Parkinson’s Disease).
William S. Knowles developed industrial-scale catalytic asymmetric hydrogenation, notably for the synthesis of L-DOPA. It includes the uses of chiral diphosphine ligands (like DiPAMP) with rhodium catalyst to create a chiral environment for hydrogenation. Di-PAMP comprises two 2-methoxyphenylphenylphosphino groups bridged by an ethane group.
B. Biocatalysis (The Nature Approach)
Using enzymes (like Lipases or Reductases). Since enzymes are made of L-amino acids, they are inherently chiral “pockets.”
Example: Synthesis of Pregabalin (Lyrica)
Pregabalin is used for neuropathic pain. The industry (Pfizer) shifted from traditional synthesis to an enzymatic route.
- The Enzyme: Lipase (specifically Thermomyces lanuginosus lipase).
The Action: The enzyme “looks” at a racemic mixture of an intermediate and only reacts with one enantiomer, leaving the other untouched (Kinetic Resolution). This is highly efficient and “Green” because it happens in water at room temperature.
Real-Life “High Stakes” Examples
The Sweet and the Bitter (Taste Receptors)
- L-Asparagine: Tastes bitter.
- D-Asparagine: Tastes sweet. The chemical formula is identical, but your tongue is a chiral sensor that perceives them differently.
The Life-Saving Difference: Ethambutol
- (S,S)-Ethambutol: A powerful drug used to treat Tuberculosis.
- (R,R)-Ethambutol: Does not treat TB and actually causes blindness (optic neuritis).
- Why it matters: As a pharmaceutical chemist, if your synthesis isn’t “Absolute” or “Partial” enough, your medicine could accidentally cause permanent disability.
Comparison Table for Quick Reference