Understanding conformational isomerism is like learning the “body language” of molecules. It is about understanding how a drug ‘speaks’ to a receptor. By mastering how Ethane, Butane, and Cyclohexane twist and turn, you gain the power to predict how drug molecules behave in the human body. Whether it is the staggered stability of Ethane or the specific ‘Gauche’ fit of Acetylcholine, the 3D shape of a molecule defines its destiny in the human body. As future Pharmacists, remember: If the shape isn’t right, the medicine won’t fight.
Definition
Conformational isomers (also known as conformers or rotamers) are a type of stereoisomerism where isomers can be interconverted exclusively by rotation about single (σ) bonds. Conformers are temporary shapes; Isomers are permanent structures (unless bonds are broken). “Rotamer” is simply a shorter name and it stands for “Rotational Isomer.” The stability of a conformation is determined by the absence of Torsional, Steric, and Angle strain.
The Three “Golden Rules” of Conformers:
- No Bond Breaking: You never break a chemical bond to change a conformation; you only “twist” it.
- Identical Connectivity: The atoms are connected in the exact same order (unlike structural isomers).
- Energy Barriers: While these rotations happen millions of times per second at room temperature, there is always a small energy cost (strain) associated with the rotation.
Suitable Presentations (The “Visual Language”)
To represent these 3D shapes on a 2D page, organic chemists use three primary “presentations.” You must be comfortable drawing all three.
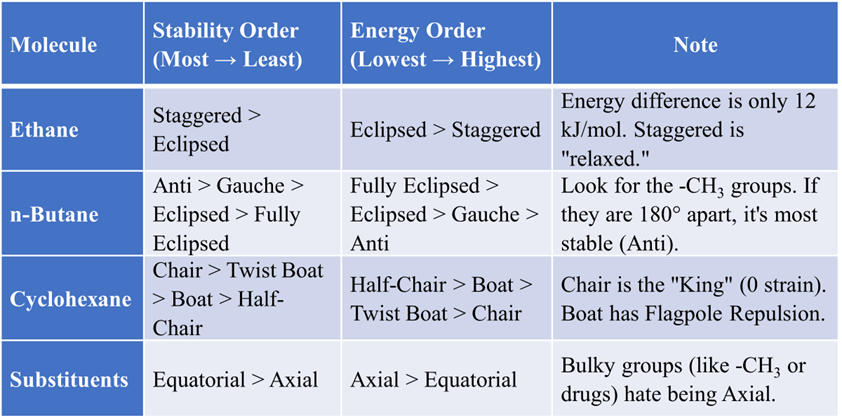
Newman Projections
Drawing a 3D molecule on a 2D chalkboard is hard. In 1952, Melvin Newman gave us a “hack” called the Newman Projection. This is the most common way to study Ethane and Butane. You look directly down the axis of a specific carbon-carbon bond.
- Front Carbon: Represented by a dot in the center.
- Back Carbon: Represented by a large circle.
- Utility: Excellent for visualizing Torsional Strain and Dihedral Angles.
Dihedral Angle: This is the angle between a bond on the front carbon and a bond on the back carbon. It’s the “angle of the twist.”
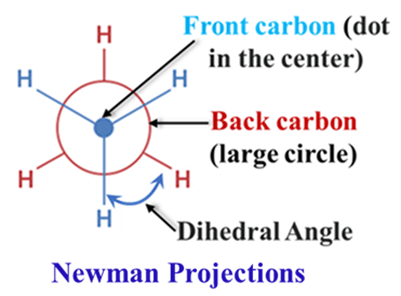
Sawhorse Projections
This view looks at the C-C bond from an oblique (side-on) angle. It looks like a “sawhorse” used in carpentry.

A sawhorse is named for its function and shape, acting as a “horse” (a support structure or frame) used to hold wood while it is being cut with a “saw.” It is often in pairs, to keep the material steady and off the ground. In historical terms, a “horse” was used to describe various devices that acted as a support or base. The four-legged frame mimics a horse, supporting a load.
- Lines: The C-C bond is drawn as a long, slanted line.
- Utility: Good for showing the spatial relationship between all atoms at once, often used in textbooks to show the “transition” between shapes.
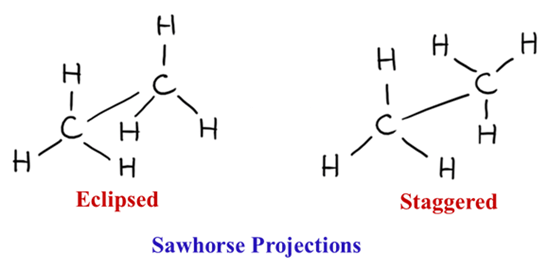
Wedge-and-Dash: The “3D-on-Paper” View
This is the standard way to show depth in a single molecule.
- Solid Wedge: The bond is coming toward you (out of the page).
- Dashed Line: The bond is going away from you (into the page).
- Normal Line: The bond is in the plane of the paper.
Stereo-dynamics
Here is why molecular shape (conformation) is the heartbeat of pharmacology:
The “Lock and Key” Hypothesis
Think of the biological receptor (a protein or enzyme) as a high-security lock. The drug is the key.
- A key is made of the right material (functional groups), but if the “teeth” of the key aren’t aligned correctly (conformation), the lock won’t turn.
- Because receptors are three-dimensional, they have specific “pockets.” Only a drug molecule in a specific conformation can tuck into those pockets to trigger a biological response.
The “Active Conformation”
A molecule like n-Butane or a complex drug isn’t static; it’s constantly twisting.
- Global Minimum: The most stable shape the drug takes while floating in the blood (e.g., the Anti form).
- Active Conformation: The specific shape the drug must “twist” into to fit the receptor.
- The Energy Cost: If a drug has to spend too much energy to twist into its active shape, it will be a weak drug. Pharmaceutical scientists try to design drugs that are “pre-organized” into their active shapes.
Example: Many neurotransmitters must adopt a specific “Gauche” or “Anti” orientation to cross the blood-brain barrier and bind to neurons. Sometimes, one conformation of a drug fits the “healing” receptor, while a different shape of the same molecule fits a “toxic” receptor elsewhere in the body. By understanding conformational isomerism, pharmacists can predict and minimize side effects by favoring the shape that only fits the intended target.
Conformational isomerism in Ethane (CH3-CH3)
In ethane, the two carbon atoms are linked by a sigma (σ) bond. Because this bond is cylindrical, the CH3 groups can rotate freely. Think of a C-C bond like an axle on a car. The atoms attached to these carbons can rotate around that axle almost freely. However, they aren’t “free” from energy costs. As the atoms spin, they pass through different arrangements called Conformers.
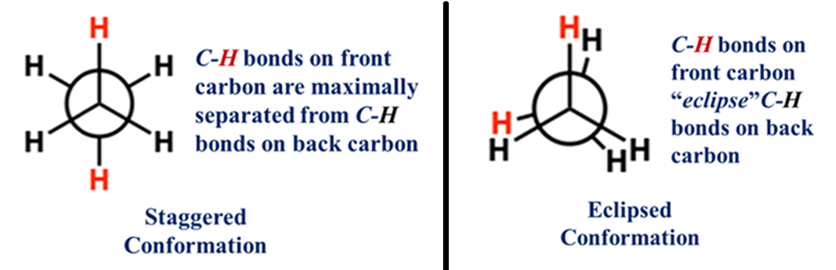
Staggered Conformation
The hydrogens are as far apart as possible (dihedral angle, ϕ = 60°). This is the “chill” state—lowest energy, maximum stability.
Eclipsed Conformation
As the molecule rotates by 60°, the front hydrogens perfectly “hide” the back hydrogens (ϕ = 0°). The hydrogens are lined up like a solar eclipse. They repel each other, creating Torsional Strain.
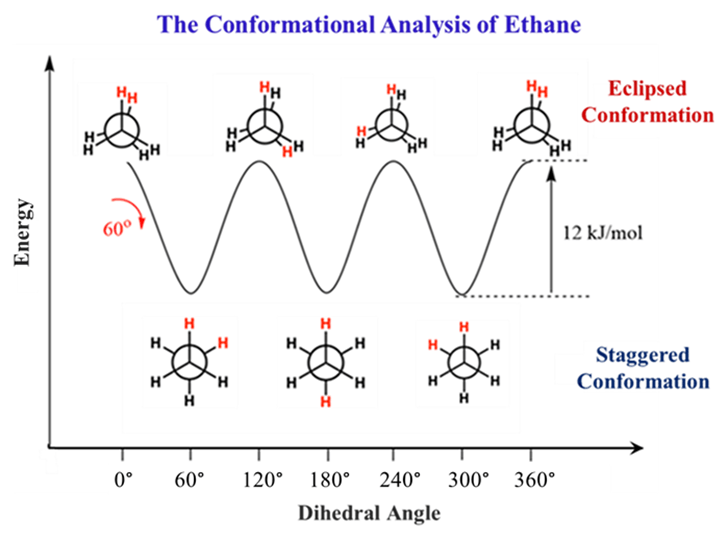
- Energy Note: The energy difference is about 12 kJ/mol. There are three pairs of eclipsed C-H bonds in the eclipsed form. Each pair contributes roughly 4 kJ/mol of torsional strain (3 × 4 = 12)
- Lowest energy, maximum stability. It’s enough to make the staggered form the preferred “home” for the molecule.
What is Torsional Strain?
It is the repulsion between the bonding electrons of the C-H bonds on the front carbon and the C-H bonds on the back carbon. Imagine two magnets with the same poles facing each other. When they “eclipse” (line up), they push away. To find peace, the molecule twists back to the staggered position where the electron clouds are furthest apart.
Stability: Staggered > Eclipsed.
Conformational isomerism in n-Butane (CH3-CH2-CH2-CH3)
Moving from Ethane to n-Butane is like moving from a small car to a SUV. In Ethane, we only dealt with tiny Hydrogen atoms. In n-Butane, we have two bulky Methyl groups that act like two people with oversized backpacks trying to pass each other in a narrow hallway. This introduces Steric Hindrance (the “don’t touch me” effect). Steric hindrance is the physical repulsion felt when atoms or groups get so close that their electron clouds overlap. Let’s look down the C2-C3 bond to see how these big groups interact. As we rotate the C2-C3 bond by 60° increments, we encounter four distinct “poses”:
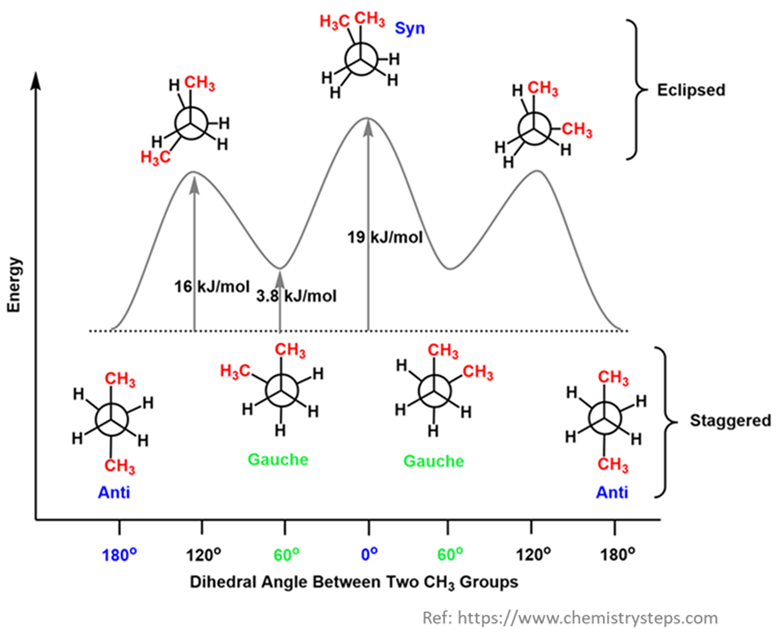
Anti (Staggered) Conformation (180°)
The two methyl groups are as far apart as possible (180° apart). Zero steric strain and zero torsional strain. Lowest energy and most stable.
Gauche (Staggered) Conformation (60°)
The Methyl groups are staggered but close to each other (60° apart). Even though they are staggered, the bulky CH3 groups are close enough that their electron clouds start to repel. This is called Steric Strain (or Van der Waals strain). Stable, but slightly “uncomfortable” due to proximity.
Eclipsed Conformation (120°)
A Methyl group on the front carbon aligns with a Hydrogen on the back carbon (overlapping). High Torsional strain and some Steric strain.
Fully Eclipsed Conformation (0°)
The two giant CH3 groups are right on top of each other. Least stable/Highest energy. This is the “Car Crash” of conformations. You have maximum Torsional strain and maximum Steric strain.
How to Draw a Newman Projection (n-Butane)
Newman projections are all about the circle and the dot.
- The Skeleton: Draw a large circle. Put a dot exactly in the center.
- Front Carbon (The Dot): Draw three lines (a “Y” shape) starting from the center dot.
- Back Carbon (The Circle): Draw three lines starting from the edge of the circle (not the dot!).
- Adding Groups:
- For Anti, put one CH3 at the very top of the front and the very bottom of the back.
- For Gauche, put them at a 60°angle (side-by-side).
- For Eclipsed, draw the back lines slightly skewed so they are “peeking” from behind the front lines.
Conformational isomerism in Cyclohexane (C6H12)
This is the “Rockstar” of the organic chemistry because many drugs have six-membered rings. Cyclohexane isn’t flat; it twists to avoid “Angle Strain.” The study of cyclohexane involves Ring Strain and the unique positions of substituents known as Axial and Equatorial. In cyclohexane-based drugs (like certain steroids or carbohydrates), each carbon has two types of bonds:
Axial (a): Six bonds pointing vertically (straight up or down).
Equatorial (e): Six bonds pointing outward (around the “equator”).
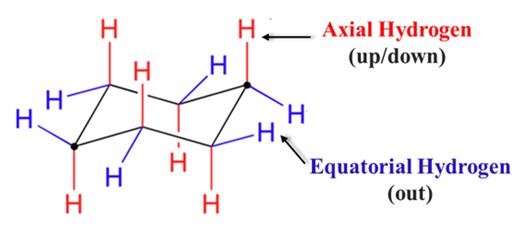
If the receptor needs the group to be Equatorial to “grab” it, but the drug is stuck in an Axial position, the drug’s potency drops significantly.
Key Pharmaceutical Concept: When a bulky group (like a drug side chain) is in an Axial position, it experiences 1,3-diaxial interactions. It physically bumps into the other axial hydrogens on the same side of the ring. Therefore, large groups always prefer the Equatorial position for stability.
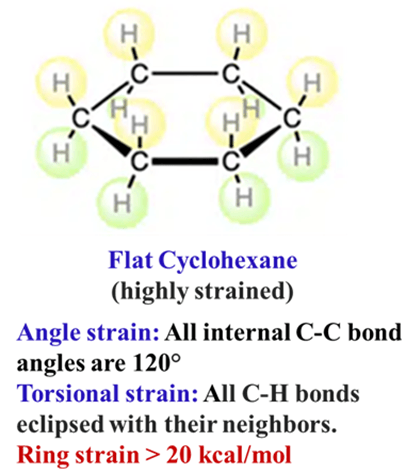
Why Cyclohexane is Not Flat?
In nature, molecules are like people—they always seek the most comfortable “posture” to avoid stress. If cyclohexane were a flat, regular hexagon, it would suffer from two major types of “molecular stress” (strain). Here is the breakdown:
Angle Strain (Baeyer Strain)
The carbon atoms in cyclohexane are sp3 hybridized. For maximum stability, these carbons want their bond angles to be exactly 109.5° (the tetrahedral angle).
- The Flat Problem: In a flat, regular hexagon, the internal angles are 120°.
- The Stress: To stay flat, the carbon bonds would have to be “stretched” out by more than 10°. This creates Angle Strain, making the flat molecule very unstable.
Torsional Strain (Pitzer Strain)
This is often the “hidden” reason.
- The Flat Problem: If the ring were flat, all twelve C-H bonds would be perfectly eclipsed with their neighbors.
- The Stress: As we learned with Ethane, eclipsing bonds create massive repulsion between electron clouds. In a flat cyclohexane, this repulsion would happen at all six carbon-carbon bonds simultaneously.
The Principal Conformations: “Puckering”
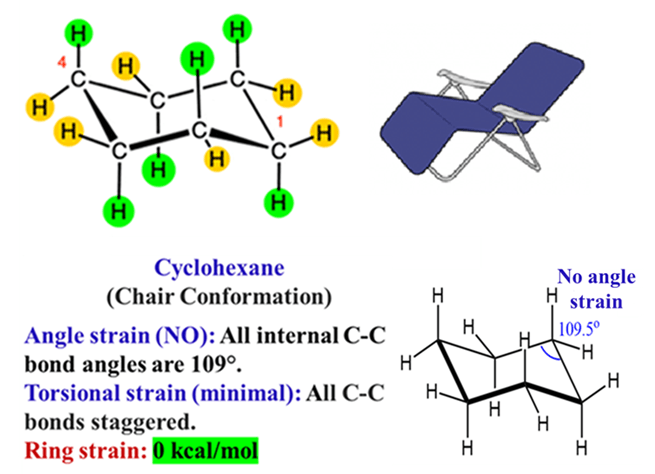
To escape these two stresses, cyclohexane “puckers” or twists out of the 2D plane into a 3D shape—the Chair Conformation.
The Chair Form (Most Stable)
- Zero Angle Strain: The bond angles shift to approximately 111°, which is much closer to the ideal 109.5°.
- Zero Torsional Strain: Every single C-C bond is in a perfectly staggered arrangement. It reduces torsional strain to nearly zero.
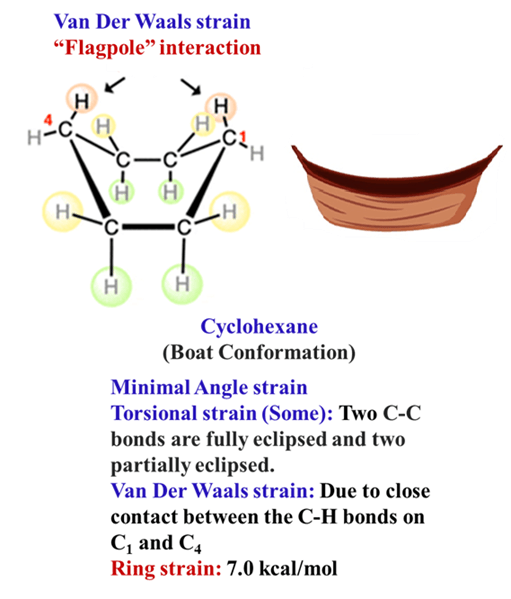
The Boat Form
High energy. By flipping one end of the chair up, you get the boat. It is significantly less stable (29 kJ/mol higher energy) due to:
- Flagpole Interactions: The hydrogens at C1 and C4 point inward and physically bump into each other.
- Torsional Strain: The bonds along the “sides” of the boat are eclipsed.
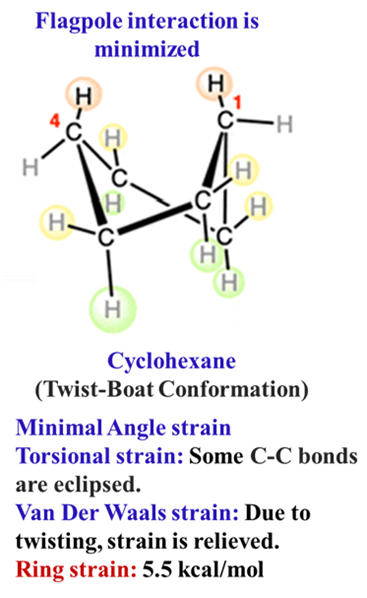
The Twist Boat
To relieve the flagpole strain, the boat twists slightly. It is a local minimum, more stable than the boat but less stable than the chair.
The Half-Chair (Transition State)
This is the highest energy state. It occurs when the molecule is midway through flipping from one chair to another. Five carbons are forced into a flat plane, causing massive strain.
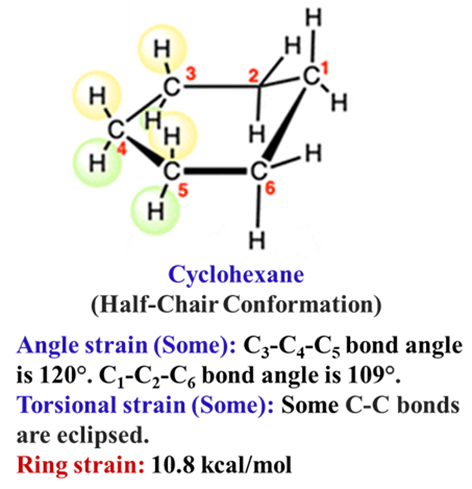
Newmann projection of cyclohexane
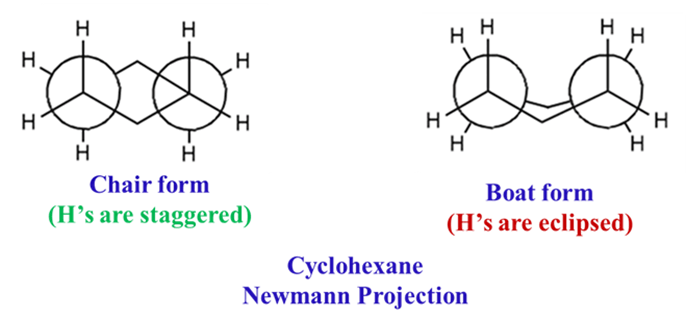
Stability & Energy of Conformations