Definition
In simple terms, a Heterocyclic compound is a cyclic organic compound in which one or more carbon atoms within the ring are replaced by heteroatoms such as Nitrogen (N), Oxygen (O), or Sulfur (S).
- Hetero = Different
- Cyclic = Ring
- Heteroatom = The “different” atom (usually Nitrogen-N, Oxygen-O, or Sulfur-S). It must be a part of the ring backbone.
Why Heterocycles Rule Pharmacy?
Heterocyclic compounds form the backbone of modern medicinal chemistry, playing a crucial role in the structure and function of numerous pharmaceutical agents. Heterocyclic compounds are frequently abundant in plants and animal products. They are one of the important constituents of almost one half of the natural organic compounds. Alkaloids, hormones, essential amino acids, hemoglobin, vitamins, natural dyes, drugs, proteins, enzymes etc. are some important classes of natural heterocyclic compounds.
- Bioisosterism: Chemists can swap a Carbon atom for a Nitrogen or Oxygen to change how a drug dissolves in the body (solubility) or how long it stays active (metabolism).
- Binding Sites: Heteroatoms like Nitrogen and Oxygen are excellent at forming Hydrogen bonds with proteins, making the drug-receptor fit much “tighter.”
- Molecular Geometry: These rings provide a rigid 3D shape, ensuring the drug hits the target like a key in a lock.
Classification of Heterocyclic Compounds
Heterocycles are classified based on different features:
Based on Ring Size
The size of the ring dictates the bond angle strain. The focus is on the “stable” rings that can survive the metabolic environment of the human body.
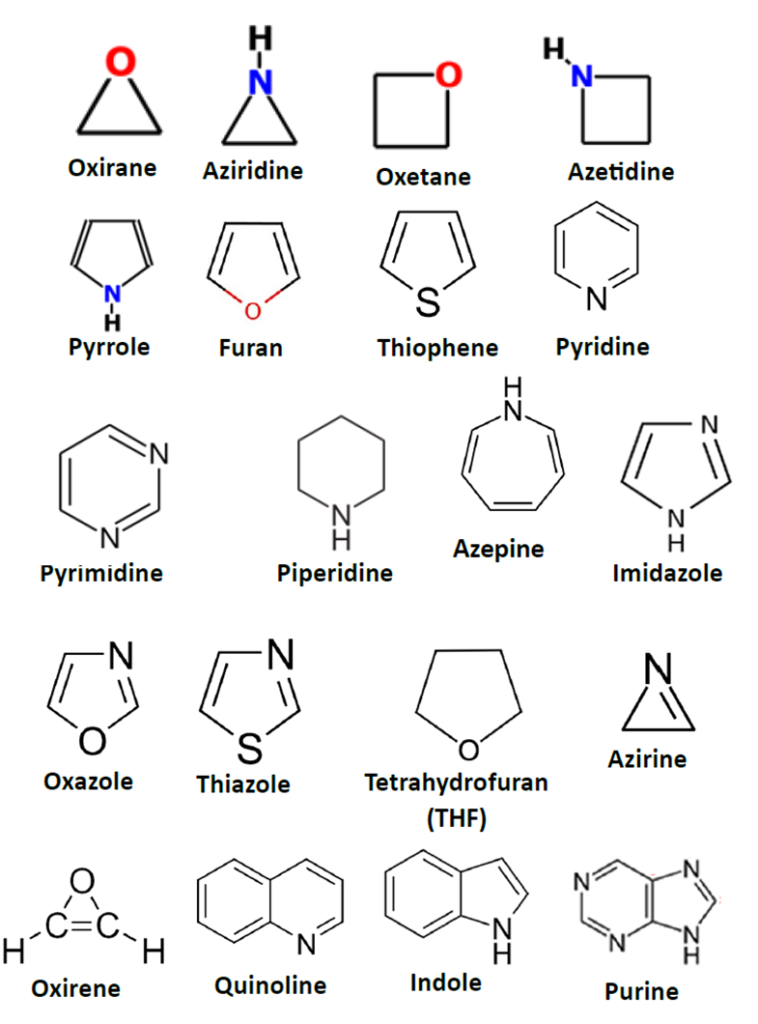
- Three-Membered Rings: High ring strain, very reactive. Examples: Oxirane (Ethylene oxide), Aziridine.
- Four-Membered Rings: Also strained; famously found in the β-lactam ring of Penicillins. Examples: Oxetane, Azetidine.
- Five-Membered Rings: Extremely common and stable. Examples: Pyrrole, Furan, Thiophene.
- Six-Membered Rings: The most stable “benchmarks” of medicinal chemistry. Examples: Pyridine, Pyrimidine, Piperidine.
- Seven-Membered Rings: Found in specialized CNS drugs. Example: Azepine (in Carbamazepine).
Based on the Nature of the Heteroatom
This is the most functional way to group them, as the heteroatom dictates the drug’s solubility and binding.
- Nitrogen-Containing: These often act as bases and can be protonated. (Pyridine, Imidazole).
- Oxygen-Containing: Usually increases water solubility and hydrogen bonding. (Furan, Oxazole).
- Sulfur-Containing: Often increases the “lipophilicity” (fat-solubility) of a drug. (Thiophene, Thiazole).
Based on Saturation (Electronic Structure)
This is a critical exam topic because it determines Aromaticity.
- Saturated Heterocycles (Aliphatic): No double bonds. They behave like open-chain ethers or amines. They are often non-aromatic. Example: Tetrahydrofuran (THF), Piperidine.
- Unsaturated Heterocycles: Contain one or more double bonds but are not necessarily aromatic. Example: Azirine, Oxirene
- Aromatic Heterocycles: These follow Huckel’s Rule (4n+2π electrons). They are very stable and flat. Example: Pyrrole, Pyridine.
Based on Molecular Architecture
- Monocyclic: A single isolated ring (e.g., Pyridine).
- Fused (Condensed) Heterocycles: Two or more rings joined together sharing a common bond.
- Benzo-fused: A benzene ring attached to a heterocycle. (e.g., Quinoline, Indole).
- Hetero-fused: Two heterocycles joined together. (e.g., Purine—found in DNA).
Structures of Heterocyclic Compounds