We’re stepping into the role of molecular detectives. In Pharmaceutical Organic Chemistry, we often encounter molecules that look identical on paper—same atoms, same bonds—yet they act like complete strangers in a biological system. We aren’t just looking at Maleic acid anymore; we’re looking at why Cisplatin kills cancer cells while its trans-counterpart does virtually nothing. Let’s get into the “how” and the “why” of configuration determination. As future pharmacists, your job isn’t just to memorize these structures; it’s to understand the analytical ‘logic’ we use to prove which one is which. Let’s break down the methods of determination of configuration of geometrical isomers.
Determining Configuration
Geometrical isomers have different physical and chemical properties because the spatial arrangement of their atoms differs. Here is how we distinguish them:
Physical Methods
A. Dipole Moment (μ) Analysis
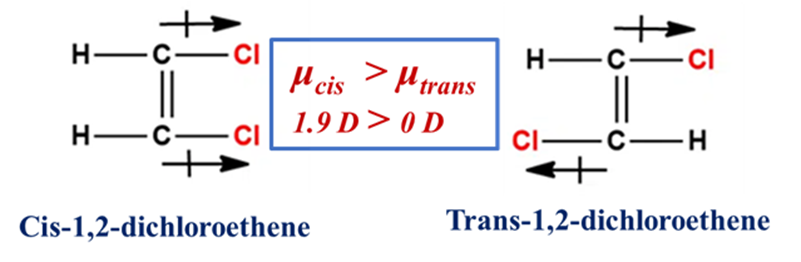
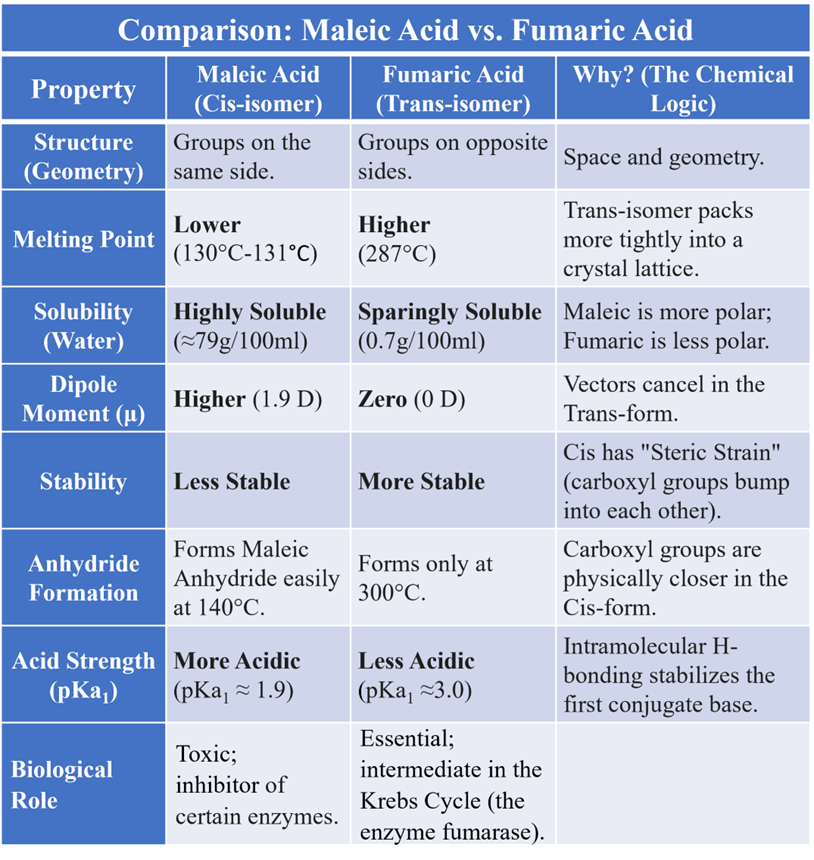
Dipole moment is a quantitative measure of the separation of positive and negative electrical charges within a system. It represents the overall polarity of a molecule. It is a vector quantity, meaning it has both magnitude and direction, typically pointing from the positive charge center to the negative charge center. This is often the most reliable physical method. In a Cis-isomer, the individual bond dipoles usually reinforce each other (add up), leading to a higher net dipole moment. In a Trans-isomer, the bond dipoles often point in opposite directions and cancel out. The rule of thumb is: μcis > μtrans. This affects how the molecule interacts with solvents and drug receptors.
Example: Cis-1,2-dichloroethene has a significant dipole moment, while the Trans-isomer is nearly zero because the C-Cl vectors cancel each other.
B. Melting Point (Symmetry vs. Stability)
Generally, Trans-isomers have higher melting points. Why? Because they are more symmetrical and “pack” more tightly into a crystal lattice. It requires more energy to break.
C. Boiling Point (Polarity effects)
Usually, Cis-isomers have higher boiling points because they are more polar (higher dipole moment), leading to stronger intermolecular dipole-dipole attractions.
D. Solubility
Cis-isomers generally have higher solubility in polar solvents because they are more polar themselves. Trans-isomers, being more “compact” and symmetrical in their crystal form, are harder for solvent molecules to break apart.
Chemical Method
Method of Cyclization (Anhydride formation)
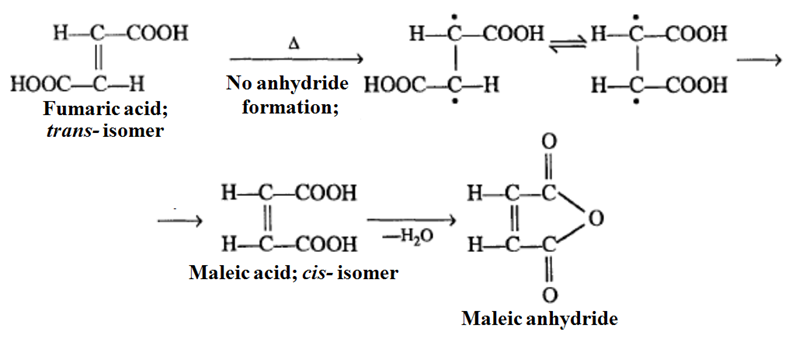
This is a classic “proximity” test. If a dicarboxylic acid can easily lose water to form an anhydride, the two -COOH groups must be on the same side (Cis).
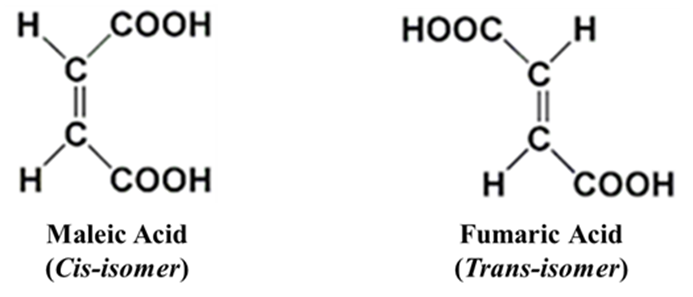
Maleic Acid vs. Fumaric Acid
Maleic Acid (Cis): The two -COOH groups are close together. It easily removes water to form Maleic Anhydride at 140 °C.
Fumaric Acid (Trans): The groups are too far apart. It does not form an anhydride easily. On strong heating (at 300°C) the fumaric acid, high temperature ruptures π-bond. The rotation of the carboxyl groups occurs followed by formation of the π-bond and loss of water. Thus, fumaric acid gives maleic acid which on hydrolysis gives maleic anhydride.
Spectroscopic Methods: The “Digital Fingerprint”
While physical properties like melting points are great for the lab, Spectroscopy provides the structural proof. The two most important tools are IR and NMR.
A. IR Spectroscopy (Infrared)
The most diagnostic feature for geometrical isomers is the C-H out-of-plane bending vibration, which is a “fingerprint.”
- Cis: Harder to detect, usually around 670–730 cm-1. It is often broad and much weaker than the Trans peak.
- Trans: They show a very strong, sharp, and characteristic band in the range of 960–990 cm-1.
B. NMR Spectroscopy (Nuclear Magnetic Resonance)
In NMR, we look at the Coupling Constant (J). This measures the interaction between the two protons on the double bond (CH=CH).
- Cis-Protons: Their coupling constant have lower value (J ≈ 6–12 Hz).
- Trans-Protons: They are farther apart but have better orbital overlap for coupling. They have a higher value (J≈12–18 Hz).
Note: Think “T for Tall”: Trans has the Taller (higher) number (12–18 Hz).
C. UV Spectroscopy
Trans-isomers are generally more planar than Cis-isomers. This planarity allows for better π -orbitals to overlap more effectively (conjugation). It lowers the energy required for electronic transitions, resulting in a shift toward a longer wavelength (λ max).
Pharmaceutical Case Studies: Why This Matters?
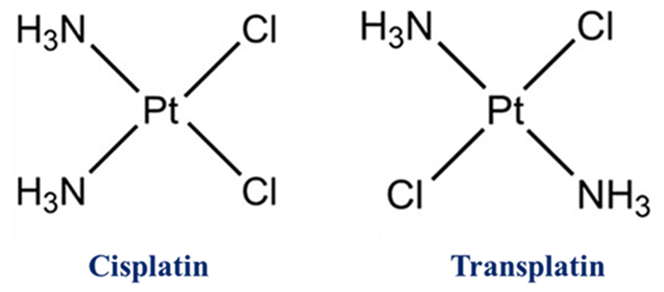
Case A: Cisplatin vs. Transplatin
This is the most famous example in medicinal chemistry.
- Cisplatin (cis-[Pt(NH3)2Cl2]): The two chlorine atoms are on the same side. This allows the molecule to bind to DNA inside a cancer cell, forming a “cross-link” that prevents the cell from dividing.
- Transplatin: Because the chlorines are opposite, it cannot form that specific cross-link with DNA, making it effectively useless as an anti-cancer drug.
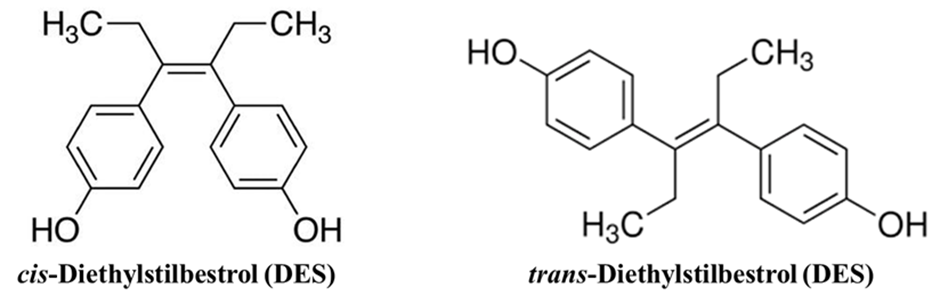
Case B: Diethylstilbestrol (DES)
DES is a synthetic estrogen.
- The Cis-isomer is only about 1/10th as active because its “bulky” groups are on the same side, preventing it from fitting into the estrogen receptor.
- The Trans-isomer is a highly potent estrogen because its shape perfectly mimics the natural hormone estradiol.
Summary
Determining the configuration of geometrical isomers is essentially a detective job. By looking at how a molecule reacts to heat (Cyclization) or how it interacts with an electric field (Dipole Moment), we can confidently label it as Cis or Trans. Mastering these spectroscopic and chemical tests is what separates a technician from a Pharmaceutical Chemist. By observing the J-values in NMR or the C-H bending in IR, you are looking at the 3D shape of a molecule that could eventually become a life-saving medication. Understanding these “molecular personalities” is vital for predicting how drugs will interact with receptors in the human body.
Comparison: Maleic Acid vs. Fumaric Acid
Laboratory Challenge: Identify the Isomer
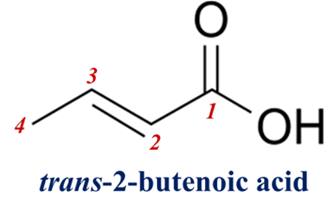
Problem: A student is given a sample of 2-Butenoic acid (an intermediate in fatty acid metabolism). The lab report provides the following data:
- IR Spectrum: A sharp, strong peak at 970 cm⁻¹.
- ¹H-NMR Spectrum: The vinylic protons appear as a doublet of quartets with a coupling constant (J) of 15.5 Hz.
Identify the isomer and justify your answer.
Solution:
- IR Clue: The peak at 970 cm⁻¹ is the signature “fingerprint” of a Trans (E) isomer (C-H out-of-plane bending).
- NMR Clue: A J-value of 15.5 Hz falls perfectly within the 12–18 Hz range characteristic of Trans protons.
- Conclusion: This acid is Trans-2-butenoic acid.