Introduction: Why Should Pharmacy Care?
In pharmacy, we are not just dealing with chemicals — we are dealing with molecules that interact with the human body. As future pharmacists, understanding the “personality” of molecules is vital. You already know that many drug molecules are chiral, meaning they exist in two mirror-image forms called enantiomers. Just like our hands look similar but cannot be superimposed, enantiomers behave differently in the body.
Sometimes:
- One enantiomer is therapeutic
- The other may be inactive or even harmful
That is why understanding racemic modification and their resolution (how we pull them apart) is extremely important.
Racemic Modification (The Racemate)
Definition
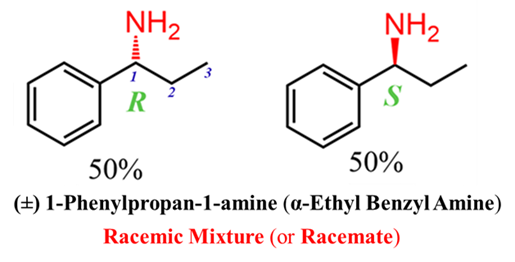
A Racemic Modification (or Racemate) is an equimolar mixture of 50:50 blend of two enantiomers. It contains equal amounts of the (+)/ d and (-) l forms.
Key Characteristics:
- Optical Activity: It is optically inactive. Because the rotation caused by one enantiomer is exactly canceled out by the equal and opposite rotation of the other, the net rotation is zero. This is called external compensation.
- Notation: We denote it using the prefix (±) or (dl). For example, (±)-Tartaric acid.
Physical States of Racemates:
When enantiomers crystallize together, they can form three types of solids:
- Racemic Mixture (Conglomerate): A physical mixture of two crystal types (pure D and pure L).
- Racemic Compound: The D and L molecules are arranged evenly in the same crystal lattice. This is the most common form.
- Racemic Solid Solution: The molecules are distributed randomly in the crystal without a specific ratio.
Resolution (Separation) of Racemic Mixtures
Since enantiomers have the same boiling points, melting points, and solubilities, you cannot separate them by simple distillation or filtration. Resolution is the process of separating a racemic mixture into its individual enantiomers.
Here are the primary methods we use in the pharmaceutical industry:
Mechanical Separation
This was used by Louis Pasteur. If the crystals of the (+) and (-) forms have different shapes, you can literally pick them apart using a magnifying glass and tweezers. However, this is rarely practical today!
Chemical Method (Diastereomer Formation)
This is the most common laboratory method. We react the racemic mixture with an optically active reagent (called a resolving agent).
- The Logic: Enantiomers + Chiral Agent → Diastereomers.
- Unlike enantiomers, diastereomers have different physical properties (solubility, boiling point). We separate the diastereomers (e.g., by fractional crystallization) and then chemically convert them back to the pure enantiomers.
Example: To resolve (±) Acid, we react it with a (-) Base. This forms two salts: one is (+) Acid-(-)-Base and other one is (-) Acid-(-)-Base. These are diastereomers and can be separated easily. In this process, we use (-)-Cinchonine, which is a naturally occurring, optically active alkaloid (a base).
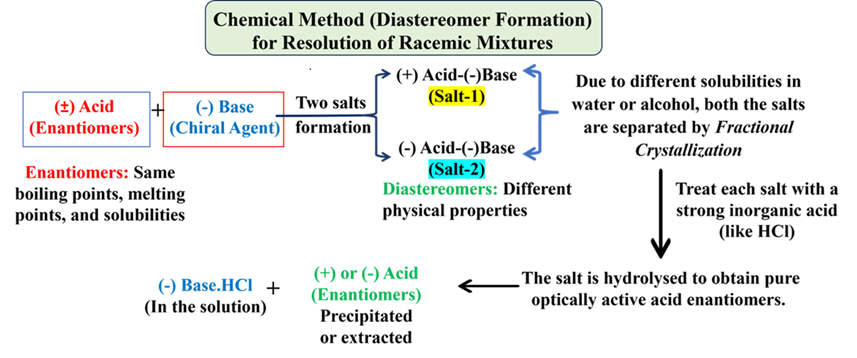
Step-by-Step Chemical Process:
When we mix a racemic mixture of Tartaric acid with a single enantiomer of a chiral base, the resulting salts are no longer mirror images—they are diastereomers.
1. The Reaction
We take the (±)-Tartaric acid and react it with a chiral base (-) Cinchonine .
2. Formation of Diastereomers
The two salts of (+) (-) and (-) (-) combinations are formed. These two salts are diastereomers. Because they are not mirror images, they have different solubilities in water or alcohol.
3. Fractional Crystallization
This is where the “magic” happens. In a solvent like water:
- (+)-Tartrate ⋅ (-) -Cinchonine is less soluble. It crystallizes out of the solution first.
- (-)-Tartrate ⋅ (-) -Cinchonine remains dissolved in the liquid (the mother liquor).
By simply filtering the mixture, you have physically separated the two components.
4. Regenerating the Pure Enantiomers
Now that we have the separated salts, we need to get rid of the Cinchonine (Base) to get our pure acid back. We treat each salt with a strong inorganic acid (like HCl or H2SO4).
(+)-Tartrate ⋅ (-) -Cinchonine + HCl → (+)Tartaric Acid + (-)Cinchonine⋅HCl
The Cinchonine hydrochloride stays in the solution, and the pure (+) or (-) Tartaric acid can be precipitated or extracted.
In the lab, always remember that the choice of the resolving agent is key. If Cinchonine doesn’t give you a large enough difference in solubility, you might try Quinine, Brucine, or Strychnine. It’s a bit like finding the right key for a lock!
Common Resolving Agents in Pharmacy
For Resolving Acidic Drugs
If your drug is an acid (like Ibuprofen), you need a chiral base to form the salt. Alkaloids are the most popular choice here because nature provides them in high optical purity.

For Resolving Basic Drugs
If your drug is a base (like many alkaloids or synthetic amines), you need an optically active acid.
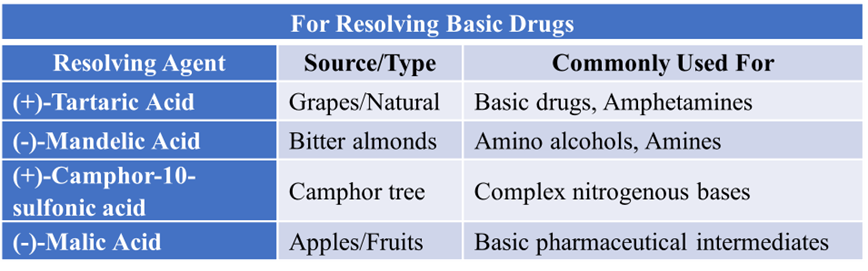
How do we choose the “Right” Agent?
Not every base works with every acid. In the industry, we look for three main criteria:
- High Enantiomeric Purity: The agent itself must be 100 % (+) or (-). If the “key” is broken, it won’t unlock the mixture.
- Solubility Difference: The two diastereomeric salts formed must have a significant difference in solubility. If both salts dissolve easily, they won’t separate by crystallization.
- Ease of Recovery: You must be able to get your drug and your (often expensive) resolving agent back easily at the end.
Note: Remember that many of these agents, like Brucine or Strychnine, are extremely toxic. In modern “Green Chemistry,” there is a huge push toward using safer, bio-based agents like L-Tartaric acid or L-Proline.
Biochemical (Enzymatic) Method
It is often referred to as Kinetic Resolution. Enzymes are protein molecules made of L-amino acids, making the enzymes themselves chiral. Because an enzyme has a uniquely shaped 3D active site, it acts like a “lock” that only fits one “key” (enantiomer) perfectly.Certain microorganisms or enzymes (like yeast or molds) are “picky eaters.” They may consume one enantiomer and leave the other untouched.
The production of L-DOPA (3,4-dihydroxyphenylalanine) is one of the most famous applications of biochemical resolution in the pharmaceutical industry. When a racemic mixture is exposed to a specific enzyme:
- The Eutomer (Preferred Isomer): Fits into the enzyme’s active site and undergoes a chemical reaction (e.g., hydrolysis, oxidation, or reduction). Example: L-DOPA (Eutomer) crosses the blood-brain barrier and is converted into dopamine to treat Parkinson’s disease.
- The Distomer (Unwanted Isomer): Does not fit and remains completely unchanged in the solution. Example: D-DOPA (Distomer) is not pharmacologically active in the same way and can lead to side effects like granulocytopenia (reduction in white blood cells).
Biochemical resolution of L-DOPA
The asymmetric hydrolysis method is used to separate the pure enantiomeric form.
Preparation
The racemic mixture is first converted into a its derivative (N-acetyl-(±)-DOPA) by acetylation reaction. Racemic DOPA is reacted with Acetic Anhydride in the presence of a base (like Sodium Hydroxide) or an acid catalyst.
The Enzyme
The enzyme Recombinant Acylase (derived from microorganisms like Aspergillus oryzae) is added.
The Selective “Clip”
The enzyme specifically recognizes the L-configuration. It “clips” (hydrolyzes) the acetyl group of the L-isomer but ignores the D-isomer.
- N-acetyl-L-DOPA + Acylase → L-DOPA (Pure Amino Acid)
- N-acetyl-L-DOPA + Acylase → D-DOPA (Unchanged)
Separation
L-DOPA is significantly less soluble at certain pH levels than its acetylated D-counterpart, allowing for easy precipitation and collection.
Added Value: “Racemization-Recycle” Technique
Industry recycle the following process to overcome the 50% yield limit of biochemical resolution.
- The unwanted N-acetyl-D-DOPA is not thrown away.
- It is treated with heat or chemicals to racemize it back into a 50:50 (±) mixture.
- This mixture is fed back into the enzyme tank.
- By repeating this cycle, the overall yield can approach 100%.
Advantages and Disadvantages

Chromatographic Method
We pass the racemic mixture through a column packed with a chiral stationary phase. One enantiomer binds more strongly to the column than the other, causing them to exit at different times.
The Core Principle: Chiral Recognition
In standard chromatography, enantiomers move at the same speed because the stationary phase is achiral. In Chiral Chromatography, we introduce a Chiral Stationary Phase (CSP).
- The “Three-Point Interaction” Model: For an enzyme or a CSP to distinguish between two enantiomers, there must be at least three points of interaction (hydrogen bonding, hydrophobic forces, or steric hindrance).
- One enantiomer (the “best fit”) binds more strongly to the CSP and moves slowly.
- The other enantiomer binds weakly and exits (eluates) the column first.
Types of Chiral Chromatography

Real-World Example: Chiral HPLC
To check the Enantiomeric Excess (ee) of a drug like Naproxen, High-Performance Liquid Chromatography (HPLC) with a chiral column is used in the industry.
- Injection: The racemic drug is injected into the HPLC.
- Differential Travel: As the mobile phase pushes the drug through the CSP, the (R) and (S) isomers separate into two distinct “peaks” on the chromatogram.
- Detection: The detector measures the area under each peak to determine the exact percentage of each enantiomer present.
Why This Matters in Pharmacy ?
In pharmacy, the “wrong” hand can be dangerous. While one enantiomer may cure a disease, the other might be toxic or inactive.
For instance, the drug Thalidomide:
- (R)-enantiomer: Safe and helps with morning sickness.
- (S)-enantiomer: Causes severe birth defects.
Interestingly, clinical studies often show that drug metabolism can vary across different populations. For example, the rate at which enzymes resolve or metabolize certain chiral drugs can differ based on genetic factors often associated with broad ethnic groupings. In the U.S., statistics show that nearly 20% to 25% of drugs currently on the market are sold as racemic mixtures, though the FDA now strongly encourages the development of single-enantiomer drugs (chiral switches) to improve safety and efficacy.
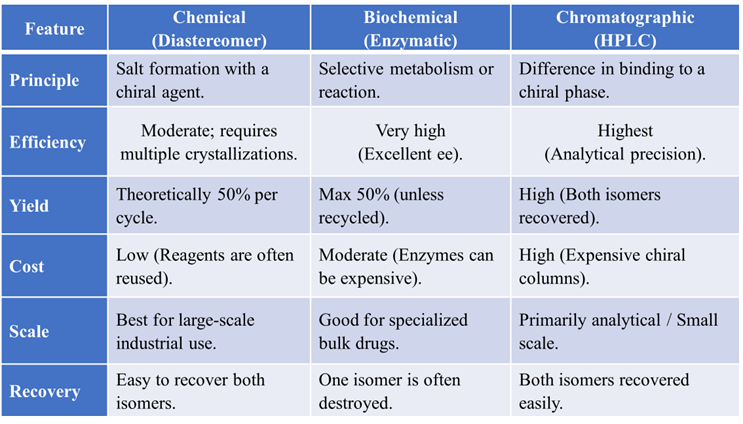
Comparison Table of Resolution Methods
Frequently Asked Questions (FAQ)
Ans: Enantiomers have identical physical properties, such as boiling points, melting points, and solubility in common solvents. Since distillation relies on a difference in boiling points to separate liquids, it is impossible to separate a racemic mixture using this method. You must first convert them into diastereomers, which do have different physical properties.
Ans: While both are optically inactive, the reason is different:
Racemic Mixture: Inactive due to external compensation (a 50:50 mix of two different molecules). It can be resolved into individual enantiomers.
Meso Compound: Inactive due to internal compensation (a single molecule has an internal plane of symmetry). It cannot be resolved because the molecule itself is achiral.
Ans: Technically, no. A racemic mixture is a 1:1 molar mixture of two different substances (the d and l enantiomers). However, in a lab setting, it often behaves like a single substance with a specific melting point (often different from the pure enantiomers) called a racemic compound.
Ans: The most widely used laboratory method is chemical resolution. This involves reacting the racemic mixture with a pure chiral reagent (like a chiral acid or base) to form a pair of diastereomeric salts. These salts are then separated by fractional crystallization based on their different solubilities.
Ans: In theory, yes. However, the ease of resolution depends on the functional groups present in the molecule. If a molecule lacks a “handle” (like a carboxylic acid, amine, or alcohol group) to react with a chiral reagent, it may require more advanced techniques like Chiral HPLC.