If you’ve ever wondered why one drug works perfectly while its “mirror image” might be toxic, the answer often lies in how these molecules are synthesized. Before we get into the technical definitions of “selective” and “specific,” we need to answer a very fundamental question: Why does the 3D shape of a molecule even matter? Till now you’ve drawn molecules on flat paper. But in the real world—and especially in the human body—molecules are 3D objects. If you change their orientation in space, you change their entire biological identity.
Why Stereochemistry is the “Soul” of Pharmacy
a) The “Lock and Key” Principle
Our bodies are made of chiral building blocks (like L-amino acids and D-sugars). This means our enzymes and receptors are “chiral locks.”
- The Analogy: Think of your right hand and a right-handed glove. They fit perfectly. Now, try putting your left hand into that same glove. It doesn’t work!
- The Drug Connection: A drug molecule must have the exact 3D “handshake” with a receptor to trigger a healing response. If the stereochemistry is wrong, the drug might just float around doing nothing, or worse, it might find the “wrong” lock.
b) The Thalidomide Story (The Ultimate Warning)
In the 1950s, a drug called Thalidomide was given to pregnant women to treat morning sickness. It was sold as a mixture of two stereoisomers:
- (R)-Thalidomide: This version was safe and effective at stopping nausea.
- (S)-Thalidomide: This “mirror image” was a potent teratogen, causing severe birth defects.
This tragedy changed the pharmaceutical world forever. Today, the Indian Pharmacopoeia (IP) and global regulatory bodies (like the FDA) demand that we know exactly which isomer we are giving to a patient.
c) Potency and Side Effects
Sometimes, one isomer is simply “better” than the other:
- Ethambutol: One isomer treats Tuberculosis; the other can cause blindness.
- Ibuprofen: Only the (S)-isomer actually relieves your pain, while the (R)-isomer is essentially “dead weight” that your body has to work to convert or clear out.
When we talk about reactions in this chapter, we aren’t just making “stuff.” We are trying to be molecular architects. We want to build the right 3D shape so that the medicine we create is safe and effective. If we can control the reaction to produce only the “good” isomer, we save lives and reduce side effects. That is the goal of studying Stereoselective and Stereospecific reactions.
Stereoselective Reactions (The “Preferred” Pathway)
Imagine you are at a buffet. You can eat anything, but you choose the pasta because it looks the most delicious and is easiest to digest. In chemistry, the reaction “chooses” the path that is energetically “cheaper” (lower energy transition state) or leads to a more stable product. In these reactions, the “choice” is usually dictated by Steric Hindrance (crowding) or Electronic Stability.
Definition
In a stereoselective reaction, a single reactant can produce two or more stereoisomeric products, but one is formed more predominantly than the other.
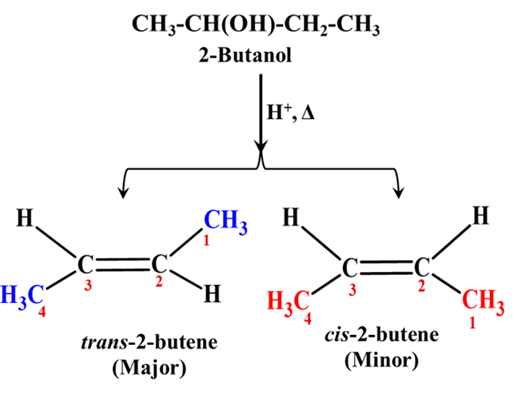
Example: Dehydration of Alcohols
Let’s look at the dehydration of 2-butanol. When we heat 2-butanol with an acid catalyst (like H2SO4), it can form two different stereoisomers of 2-butene: cis-2-butene and trans-2-butene.
- The Outcome: We get about 80% trans-2-butene and only 20% cis-2-butene.
- The “Why”: In the trans isomer, the bulky methyl (-CH3) groups are far apart, reducing “steric repulsion.” The reaction “selects” the trans version because it is more stable.
Types of Stereoselectivity
There are two “flavors” of this:
- Enantioselective: The reaction produces one enantiomer (mirror image) more than the other. (e.g., Enzyme-catalyzed reactions in the body are 100% enantioselective!)
- Diastereoselective: The reaction produces one diastereomer (non-mirror image) over the other. (e.g., The 2-butene example above).
Stereospecific Reactions (The “Fixed” Pathway)
Think of a lock and key. A specific key (reactant) only opens the door in one specific way (product). There is no “choice” involved. Imagine a train on a track. It cannot “choose” to turn left or right like a car at a fork in the road. It must go exactly where the rails (the mechanism) take it. If the rails start in the North, the train ends in the South.
Definition
In a Stereospecific Reaction, the reaction mechanism is so rigid that the stereochemistry of the starting material (reactant) completely determines the stereochemistry of the product.
If you start with a Cis reactant, you get one specific product. If you start with a Trans reactant, you get a completely different stereoisomeric product. There is no “major” or “minor” choice here. In these reactions, the mechanism is the boss.
Example
1. The SN2 Reaction
You’ve studied SN2 before, but now let’s look at its “specific” nature. Because the nucleophile must attack from the side opposite to the leaving group, the molecule’s configuration always flips. This is known as Walden Inversion.
- The Rule:
- If you start with the (S)-isomer, you will get 100% (R)-product.
- If you start with the (R)-isomer, you will get 100% (S)-product.
2. Addition of Bromine (Br2) to Alkenes
The addition of Br2 to a double bond is stereospecific because it forms a cyclic bromonium ion intermediate, forcing an “anti-addition” (the two Bromines must come from opposite sides).
- Start with Maleic Acid (Cis): The result is always a Racemic Mixture (± dibromosuccinic acid).
- Start with Fumaric Acid (Trans): The result is always a Meso Compound (meso-dibromosuccinic acid).
Why is this “Specific”? Because the Cis reactant cannot produce the Meso compound, and the Trans reactant cannot produce the Racemic mixture. The starting geometry dictates the final result.
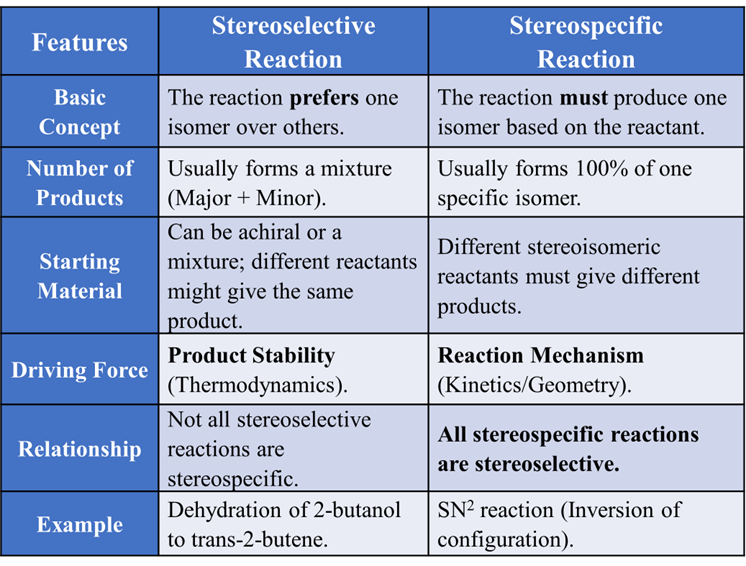
The Master Comparison Table
Difference between Stereoselective and Stereospecific reactions.
Pharmaceutical Importance (The Real World)
Why do we spend hours learning this in a Pharmacy degree? Because stereochemistry saves lives.
- Drug Safety: As we saw with Thalidomide, one isomer can be a medicine while the other is a toxin. Stereospecific synthesis ensures we only create the “safe” version.
- Drug Potency: Often, only one stereoisomer fits the biological receptor. For example, in Salbutamol (used for asthma), the (R)-isomer is a potent bronchodilator, while the (S)-isomer is inactive and may even cause side effects.
- Cost Efficiency: If a reaction is 100% stereoselective, we don’t waste chemicals making the “wrong” isomer, and we don’t have to spend money on expensive purification processes to separate them later.