Think of Atropisomerism as the chemistry of “frozen motion.” While most single bonds in organic chemistry act like spinning wheels, some bonds get “stuck” because the groups attached to them are simply too big to pass each other. This is a crucial concept because it explains how a molecule can be chiral (optically active) even if it doesn’t have a single “chiral center” (asymmetric carbon). Atropisomerism teaches us that chirality isn’t just about atoms—it’s about space. When a single bond loses its freedom to rotate, a simple molecule transforms into a pair of unique mirror images with potentially life-saving differences.
What is Atropisomerism?
The word comes from the Greek a (not) and tropos (turn).
Definition
Atropisomers (non-turning isomers) are stereoisomers that result from restricted rotation about a single bond. The “steric clash” between bulky groups creates a rotational energy barrier so high that the different shapes (conformers) can be separated and kept in bottles at room temperature. In biphenyls, this happens around the C–C single bond connecting the two phenyl rings. They are enantiomers, i.e. non-superimposable mirror images.
Example
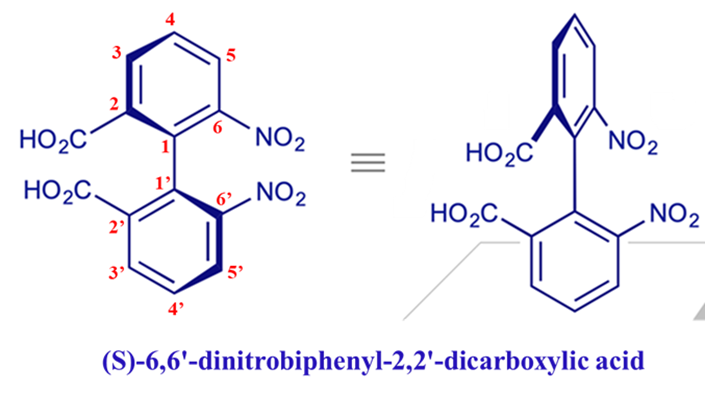
Stereo isomerism in biphenyl compounds (6,6′-dinitrobiphenyl-2,2′-dicarboxylic acid). Biphenyls with different groups at each ortho position are optically active. But biphenyls unsubstituted at both rings, or with flexible smaller groups, are optically inactive.
The “Steric Clash”
Imagine two people trying to pass each other in a very narrow hallway while carrying bulky backpacks. They might get stuck facing one direction. In biphenyls, the “backpacks” are the ortho-substituents (the groups at positions 2, 2′, 6, and 6′).
When these groups are large (like -NO2, -COOH, or -I), they bump into each other if the rings try to stay in the same plane. To find peace, the rings twist 90 degrees relative to each other. If they can’t twist back, they are “locked” into a specific mirror-image shape.
The “Why”: Restricted Rotation
Normally, a single bond allows free rotation. However, if we place large, bulky groups at the ortho positions (the positions closest to the central bond), these groups “bump” into each other. This is called steric hindrance.
To avoid this “clash,” the two rings twist out of the same plane and become perpendicular (at 90 degrees) to each other. If the rotation is blocked effectively, the molecule gets “stuck” in a twisted shape.
Conditions for Optical Activity
Only biphenyls with suitable ortho substitution and no plane of symmetry in the overall molecule show optical activity. For a biphenyl to show atropisomerism and be optically active, it must satisfy these conditions:
Axial Chirality
Unlike glucose or lactic acid, biphenyls have a “chiral axis” rather than a chiral center.
Restricted Rotation
The ortho positions must have groups bulky enough to prevent the rings from rotating 360 degrees. If the groups are too small (like H or F), the rings just spin, and the optical activity is lost.
The “Energy Wall”
The energy required to force the groups past each other must be at least 100 kJ/mol. If it’s lower, the molecule will “racemize” (flip back and forth) at room temperature.
Absence of Symmetry (A ≠ B)
This is the most important point. Each ring must be unsymmetrically substituted. If a ring has two identical ortho groups (e.g., both are -COOH groups), the molecule will have a Plane of Symmetry and will be achiral (inactive).
How to Assign R/S Configuration (The 4-Step Method)
Assigning R/S to biphenyls is challenging because it requires identifying a chiral axis rather than a traditional tetrahedral chiral center. The molecule utilizes a chiral axis defined by the central bond. Here is the easiest way to teach it:
- The Viewpoint: Look at the molecule along the central C–C bond (like looking through a telescope).
- Near vs. Far: The two groups on the ring closer to you always get higher priority (1 and 2) than the groups on the far ring (3 and 4).
- Cahn-Ingold-Prelog (CIP) Rules: Within the “near” pair, assign 1 to the higher atomic number and 2 to the lower. Do the same for the “far” pair (3 and 4).
- The Curve: Draw a path from 1 → 2 → 3.
- Clockwise = R (Rectus)
- Counter-clockwise = S (Sinister)
Stability: When does the “Flip” Happen?
Not all atropisomers are permanent. Some are stable at room temperature, while others “racemize” (lose their optical activity) over time.
- Size Matters: The larger the Van der Waals radius of the ortho groups, the more stable the isomer. (Order of stability: I > Br > Cl > F).
- Electronic Effects: Sometimes, groups that can hydrogen-bond or have electronic repulsions can further restrict or facilitate the rotation.
- Temperature: At high temperatures, the molecule gains enough “energy” to force its way through the narrow planar transition state, like a person squeezing through a very tight revolving door.
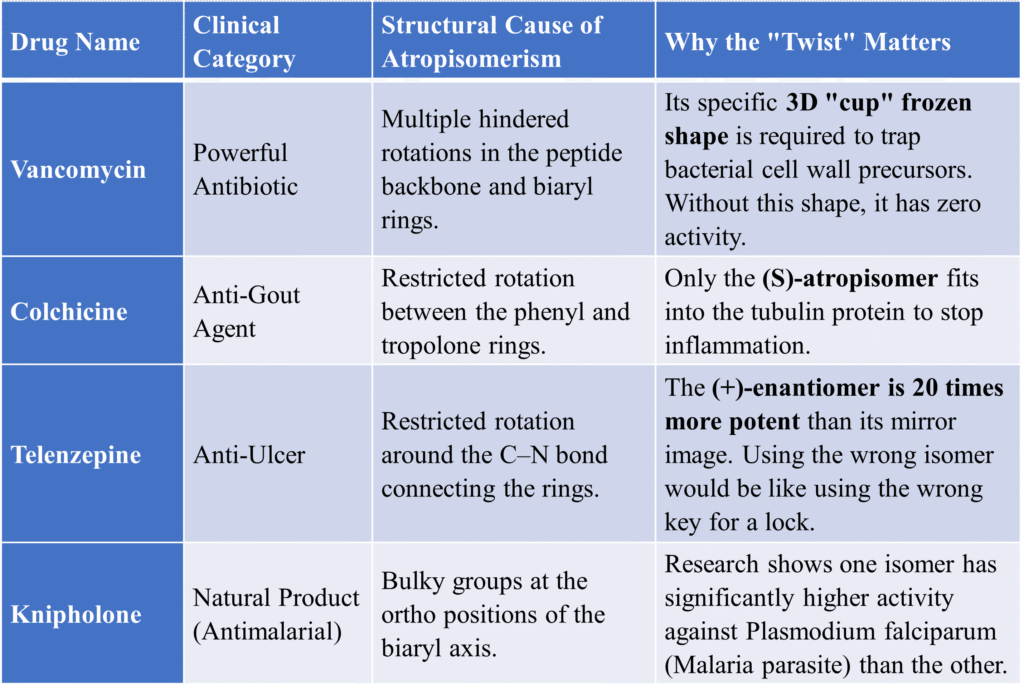
Atropisomerism in Clinical Practice
In drug design, shape is everything. If a drug molecule is an atropisomer, one “twist” might fit perfectly into a human receptor to cure a disease, while the other “twist” might be toxic or completely useless. This table highlights how “frozen” molecular shapes directly impact patient care.