Saponification is the chemical process of converting fats or oils into soap and glycerol. Chemically, it is the alkaline hydrolysis of an ester. When a triglyceride (the main component of oil) reacts with a strong base, the ester bonds are broken, releasing glycerol and creating the metal salts of fatty acids, which is known as soap.
The Chemical Reaction
The general reaction involves a triglyceride and base. Most oils like olive, coconut, or palm oil are made of “triglycerides.” The base is usually Sodium Hydroxide (NaOH) for solid soap or Potassium Hydroxide (KOH) for liquid soap. The reaction of oil and base produces soap and glycerol.
General Equation
Triglyceride + 3NaOH → Glycerol + 3 Sodium salt of fatty acid (Soap)
Specific Example: Saponification of Tristearin
When tristearin (a fat found in many animal and vegetable oils) reacts with sodium hydroxide (NaOH), it forms sodium stearate (soap) and glycerol:
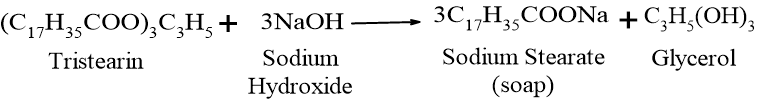
Mechanism of Saponification
The reaction occurs in three primary steps involving a nucleophilic attack on the ester group:
- 1. Nucleophilic Attack: The hydroxide ion (OH–) from the base attacks the carbonyl carbon of the ester bond in the oil, forming a tetrahedral intermediate.
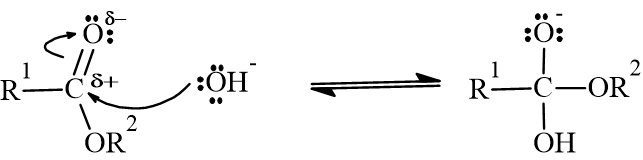
- 2. Leaving Group Removal: The intermediate collapses, breaking the bond between the oxygen and the glycerol backbone. This releases an alkoxide ion and forms a carboxylic acid.
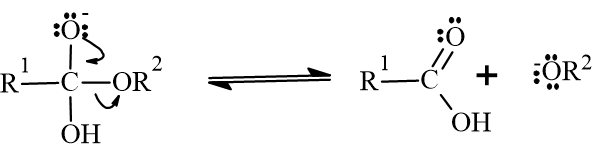
- 3. Deprotonation: The alkoxide ion (a strong base) immediately strips a proton from the carboxylic acid. This results in an alcohol (glycerol) and a carboxylate ion (the soap molecule).

Types of Soap Produced
The characteristics of the soap depend largely on the type of alkali used:
- Hard Soaps: Produced using Sodium Hydroxide (NaOH). These are typically found in solid bar soaps used for laundry and bathing.
- Soft Soaps: Produced using Potassium Hydroxide (KOH). These are softer, often liquid or semi-solid, and are used in shaving creams and liquid hand soaps.
Importance and Applications
Saponification is not just for making bathroom soap; it has several industrial and scientific uses:
- Soap and Detergent Industry: It is the primary method for manufacturing all natural soaps. It produces Glycerol as a by-product. In industrial soap making, glycerol is often removed to be sold for expensive lotions. It is used in cosmetics and pharmaceuticals. Natural soaps often feel much “creamier” because soap salts are combined with the skin-protecting power of the glycerol.
- Fire Extinguishers: Wet chemical fire extinguishers use saponification to put out grease fires. These extinguishers contain a potassium-based solution (like potassium acetate, carbonate, or citrate) mixed with water. The chemicals react with the burning oil to turn it into a non-combustible soap foam, which cut off its oxygen supply to extinguish it and cools the surface.
- Saponification Value: In food science, the “Saponification Number” is used to identify the purity of an oil or fat. It represents the milligrams of KOH required to saponify 1 gram of fat, helping determine the average molecular weight of the fatty acids present.
- Art Conservation: Interestingly, saponification can be a problem in oil paintings. An oil painting is basically made of two things the Binder (Linseed Oil) and the Pigment made from heavy metals like Lead (Lead White) or Zinc (Zinc White). Over decades, heavy metals in paint pigments act like a base and can react with the oil binder to form “metallic soaps”. This can cause visible lumps or damage to the masterpiece (art, craft, or other high-quality creation).
Summary Table
| Component | Role |
| Triglyceride | The source of fatty acids (found in oils/fats). |
| NaOH / KOH | The “chemical scissors” that break the oil apart. |
| Glycerol | The skin-softening byproduct. |
| Soap | The cleaning agent that bridges the gap between oil and water. |