Introduction:
Sachse-Mohr’s Theory was proposed by Hermann Sachse in 1890 and later refined by Ernst Mohr in 1918. It can be summarized in one sentence: “Rings don’t have to be flat.” While previous theories (like Baeyer’s) assumed that carbon rings were almost flat, Sachse and Mohr realized that larger rings “pucker” or fold into 3D shapes to stay comfortable. This theory successfully explained the stability of cyclohexane and larger rings.
Postulate:
- Non-Planar Rings (Puckering): Carbon rings with six or more members are not planar but adopt puckered (three-dimensional) conformations.
- Strain Relief: These puckered conformations allow all the C-C-C bond angles to be maintained at or very near the ideal tetrahedral angle 109.50, completely eliminating angle strain. Since angle strain is absent, these are called strainless rings.
The Core Idea: “Stop Forcing It!”
Imagine you have a long piece of stiff wire that wants to stay at a specific angle (109.5°).
- The Problem (Baeyer’s View): If you force that wire to form a flat hexagon on a table, you have to bend the angles to1200. This creates stress (strain).
- The Solution (Sachse-Mohr View): If you let the wire “pucker” up and down off the table, you can keep the wire at its favorite 109.50 angle while still forming a closed loop. There is zero angle strain.
Example: Cyclohexane (C6)
It exists primarily in two major non-planar, strainless conformations:
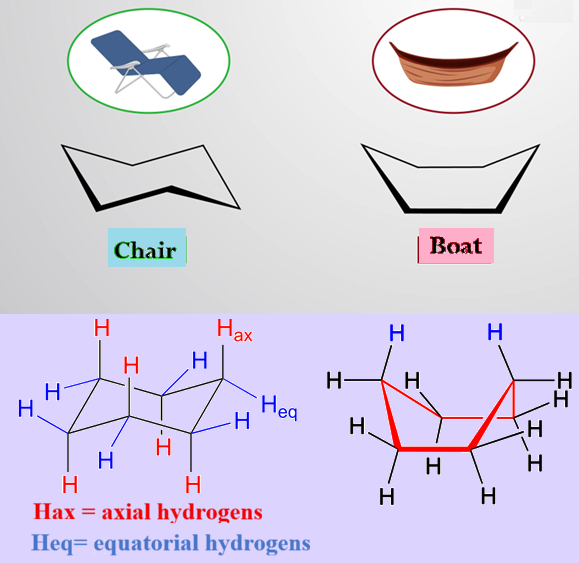
1- Chair Conformation:
It Looks like a lounge chair. It is the most “relaxed” (most stable) version where every atom has plenty of space. It eliminates both angle strain (all bond angles are 109.50) and torsional strain (all C-H bonds are staggered).
In a chair conformation, there are two types of positions for atoms to go:
i) Axial (a): These point straight up or straight down, like the axis of the Earth.
ii) Equatorial (e): These point outward toward the “equator” of the molecule.
| Quick Comparison Table | ||
| Feature | Axial Position | Equatorial Position |
| Direction | Vertical (Up/Down) | Horizontal (Outward) |
| Space | Crowded | Roomy |
| Strain | High (1,3-diaxial) | Low/Zero |
| Stability | Less Stable | More Stable |
2- Boat Conformation:
It is Less stable than the chair form. While it is free of angle strain, it possesses torsional strain (due to eclipsed C-H bonds) and steric strain (due to flagpole-flagpole H-H repulsion). It is a bit more “crowded” than the chair.
Summary
| Summary Table: Simple Comparison | ||
| Feature | Baeyer’s View (Old) | Sachse-Mohr’s View (New) |
| Ring Shape | Flat (2D) | Puckered/Folded (3D) |
| Bond Angles | Forced to change | Stay at a perfect 109.50 |
| Stability | Larger rings are unstable | Larger rings are very stable |
| Analogy | A flat sheet of paper | An origami bird |