Welcome, students! Today we are diving into one of the most important heterocyclic compounds in pharmacy: Pyridine. If you’ve ever wondered why certain drugs work the way they do, or why some molecules react while others stay quiet, understanding the “personality” of Pyridine is your first step. To understand its basicity, we must look at the Nitrogen atom’s “home.”
Introduction: Structure and Hybridization
Pyridine (C5H5N) is a six-membered heterocyclic aromatic compound.
- Hybridization: Every atom in the pyridine ring (5 Carbons and 1 Nitrogen) is sp2 hybridized.
- The Orbital Setup: Nitrogen has five valence electrons. In Pyridine:
- Two electrons form σ bonds with adjacent Carbon atoms.
- One electron sits in an unhybridized p-orbital to contribute to the aromatic 6π electron cloud.
- The Lone Pair: The remaining two electrons occupy the third sp2 hybrid orbital.
- Geometry: Because the sp2 orbital lies in the same plane as the ring (trigonal planar), it points outward, perpendicular to the π system.
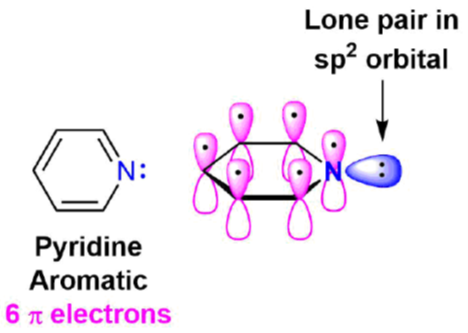
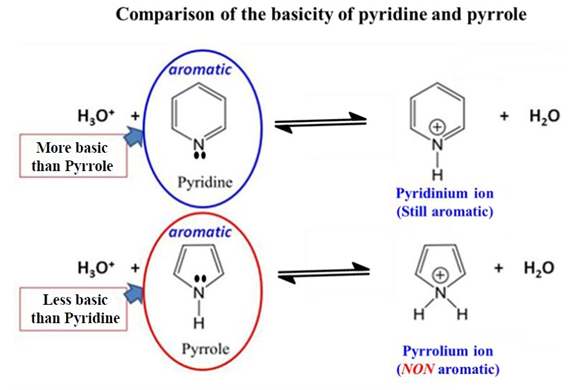
The “Lone Pair” Secret: Why Pyridine is a Base?
In organic chemistry, a base is an electron-pair donor (Lewis Base). Pyridine is a base because its nitrogen lone pair is “freely available.” It can readily “donate” to a proton (H+).
- Non-Delocalization: Unlike some other heterocycles, Pyridine’s lone pair is not involved in aromaticity. Because they are just sitting outside the ring, free and available, Pyridine is quite happy to act as a base. The ring already has 6 electrons in the p-orbitals to satisfy Huckel’s Rule (4n+2).
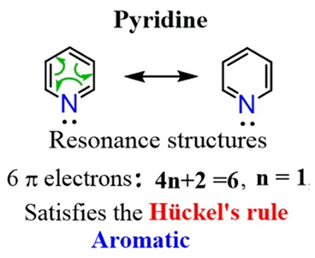
- Steric Availability: Since the lone pair points outward away from the ring atoms, it is physically accessible for a proton (H+) to approach and bond.
- Formation of Pyridinium Ion: When Pyridine acts as a base, it accepts a proton to form the Pyridinium cation. Crucially, the ion remains aromatic because the π system is left untouched.
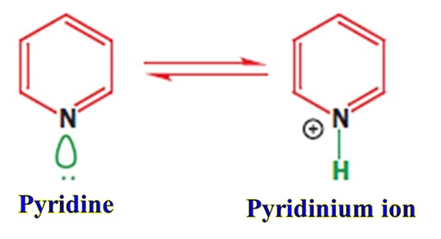
The Comparison: Pyridine vs. Others
Students often get confused here, but think of it this way:
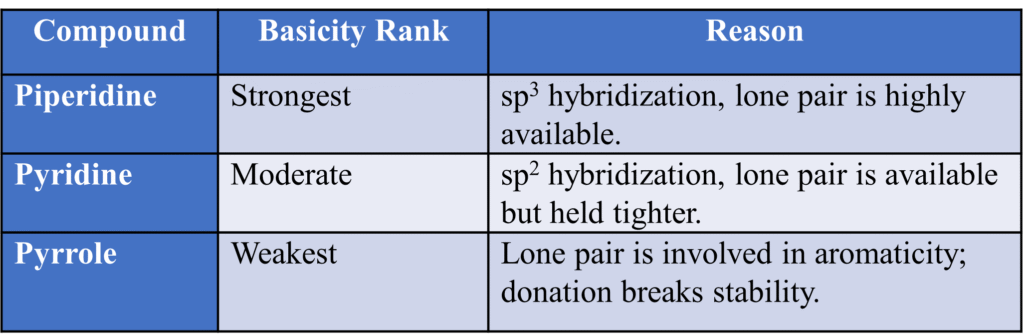
Pyridine vs. Pyrrole
- Pyrrole is a very weak base. The nitrogen’s lone pair is “busy” maintaining the aromaticity of the ring. If it gave them away to a proton, the ring would lose its aromatic stability. Therefore, Pyrrole is a very weak base. In Pyrrole, the nitrogen lone pair is part of the aromatic 6π system.
- Pyridine is a stronger base than Pyrrole because its lone pair is extra and available without breaking aromaticity.
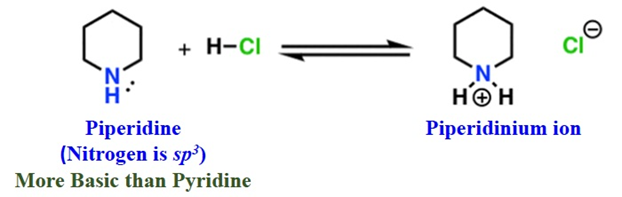
Pyridine vs. Aliphatic Amines (e.g., Piperidine)
- Pyridine is a weaker base than Piperidine.
- Reason: It comes down to s-character.
- Pyridine (Nitrogen is sp2): 33% s-character.
- Piperidine (Nitrogen is sp3): 25% s-character.
- The Logic: Electrons in sp2 orbitals are closer to the nucleus (more “s” means more “pull”) and are held more tightly. Therefore, Pyridine is less willing to share its electrons than a sp3 amine.
Resonance Effects: The Ring’s Influence
The pyridine ring is “electron-deficient” compared to benzene. Nitrogen is more electronegative than carbon, so it pulls electron density toward itself through the resonance effect.
Resonance Structures: If you draw the resonance hybrids, you will see positive charges appearing at the ortho (2, 6) and para (4) positions.
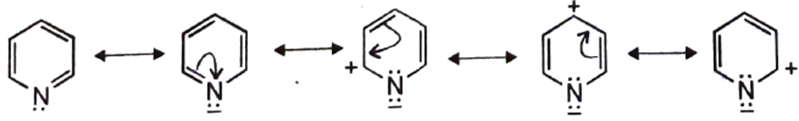
Impact on Basicity: While the ring is electron-deficient, the Nitrogen atom itself maintains high electron density. However, if you add substituents to the ring:
- EDG (Electron Donating Groups): Like -NH2 or -OCH3 at the 4-position, these increase basicity by “pushing” more density onto the Nitrogen.
- EWG (Electron Withdrawing Groups): Like -NO2 at the 4-position, these decrease basicity by “pulling” density away.
The Comparison Table: