Transitioning from basic organic chemistry to the spatial complexities of stereochemistry is often a “lightbulb moment”. It explain how a simple molecular flip can turn a potent drug into an inactive (or even toxic) substance. In the pharmaceutical world, Geometrical Isomerism isn’t just a theoretical concept—it is a critical factor in how a drug performs in the human body. When we talk about “structure-activity relationships” (SAR), the spatial arrangement of atoms can mean the difference between a cure and a chemical that does nothing (or even causes harm).
What is Geometrical Isomerism (GI)?
Geometrical isomerism (also known as Cis-Trans isomerism) is a type of stereoisomerism. The isomers have the same chemical formula and connectivity but differ in their spatial arrangement of atoms. It occurs due to restricted rotation (atoms cannot rotate freely) around a double bond or a ring structure.
Think of it like a revolving door that has been locked in place; you can be on one side or the other, but you can’t just swing through to change positions.
Essential Conditions for GI
A molecule isn’t “allowed” to show GI unless it meets two strict criteria:
Restricted Rotation
Usually due to a carbon-carbon double bond (C=C), carbon-nitrogen double bond (C=N), or a cyclic structure. Double bonds consist of a σ- and a π-bond. If there is rotation about the double bond, the π-bond will break, thus there is no free rotation about a double bond, i.e. the rotation about a double bond is frozen.
Different Substituents
Each of the two atoms involved in the double bond must be attached to two different groups.
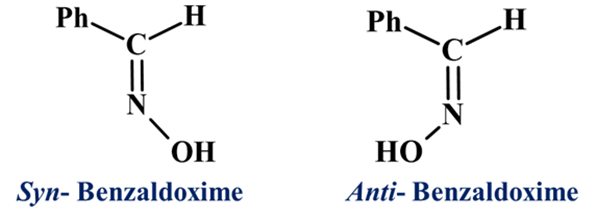
Example: Maleic Acid (Cis) and Fumaric Acid (Trans) (C4H4O4)
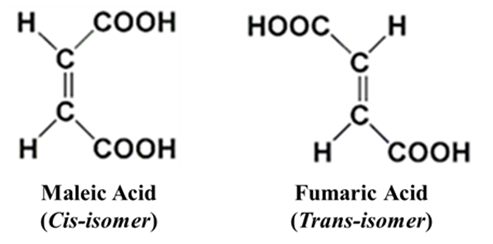
If a carbon has two Hydrogens it cannot show GI. Example: H2C=CH2 (Ethene) cannot show GI because both groups on the carbon are the same (Hydrogen).
The Nomenclature Systems
Cis-Trans System
- Cis: Identical groups are on the same side of the double bond.
- Trans: Identical groups are on opposite sides.
- Limitation: This only works well when there are two identical groups to compare.
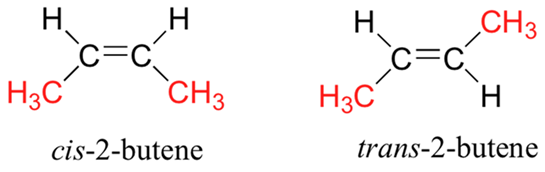
Example: In 2-butene (C4H8) the methyl groups can be located on the same side or on the opposite side of the double bond, giving rise to two geometrical isomers.
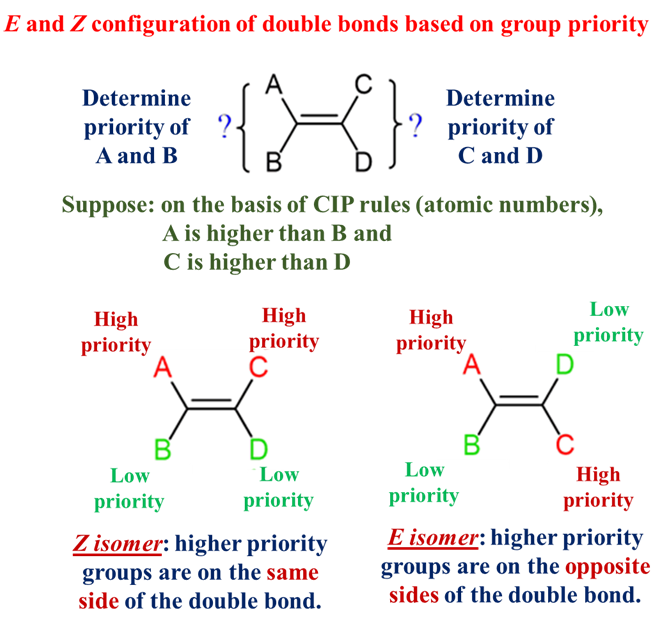
E-Z System (The Universal Method)
When a double bonded carbon atom is attached to three or four non-hydrogen substituents, cis and trans fails in such compounds. We use the E-Z system based on the Cahn-Ingold-Prelog (CIP) priority rules. The E/Z system analyzes the two substituents attached to each carbon in the double bond and assigns each either a high or low priority.
- Assign Priority: Atoms with higher atomic numbers get higher priority. Remember, if the first atoms are a “tie,” move to the next set of atoms in the chain.
- Z isomer (from German – zusammen = together): High-priority groups are on the Same side. (Think: “Zame Side”).
- E isomer (from German- entgegen = opposite): High-priority groups are on Opposite sides.
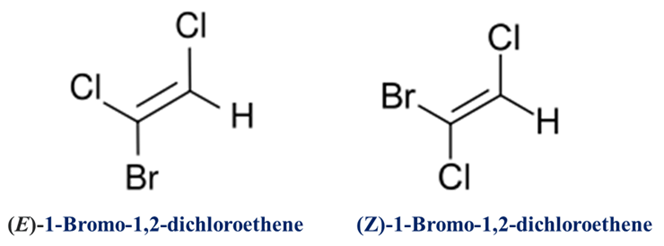
The prefixes Z and E are placed before the numbers indicating the positions of the double bonds in the main chain. It should be noted that there is no relation between cis/trans and E/Z configurations.
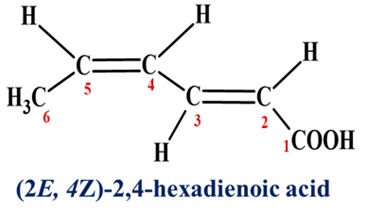
Syn-Anti System
Mainly used for Nitrogen-containing compounds like Oximes (>C=N-OH) and Azo compounds (-N=N-).
- Syn: The -OH group and the H (in aldoximes) are on the same side of the C=N double bond.
- Anti: They are on opposite sides.
- It is relevant in medicinal chemistry for understanding the stereochemistry of drugs and drug interactions.
Physical Properties of Geometrical Isomers
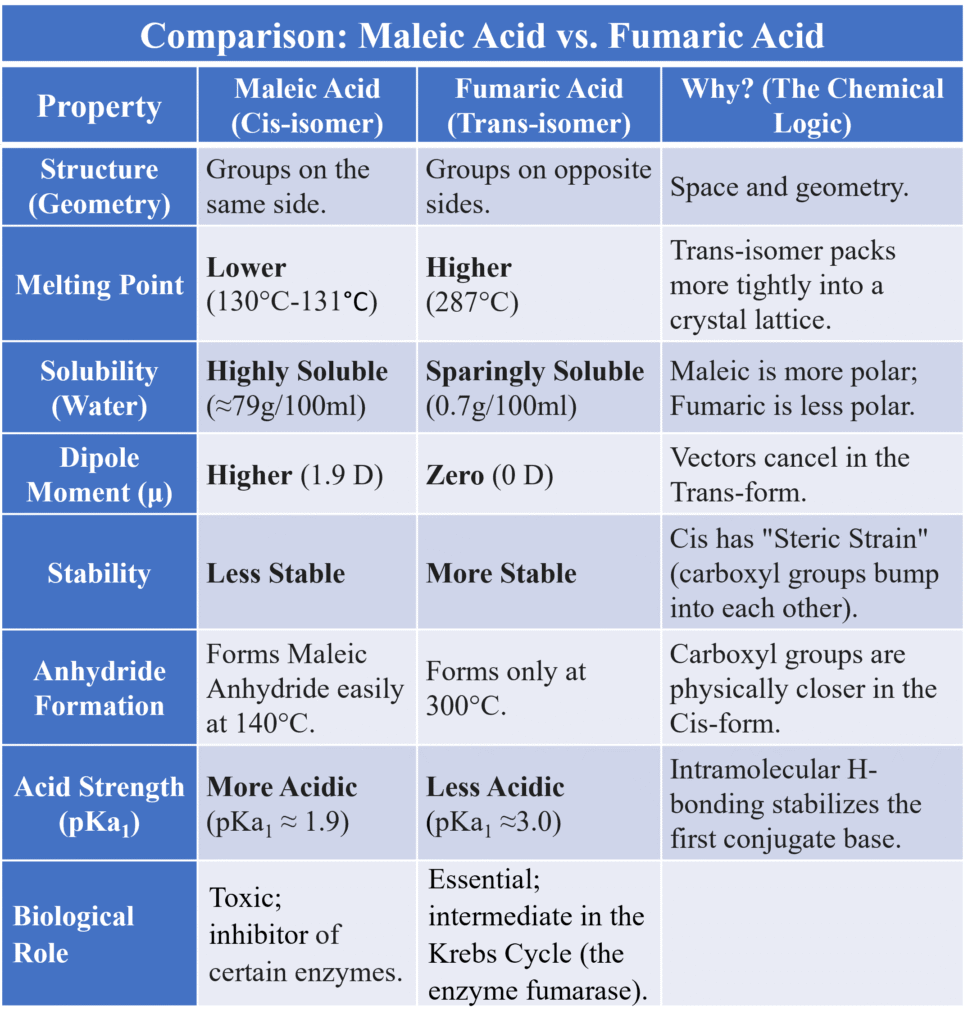
Physical properties differ significantly between Cis and Trans isomers because of their different shapes and symmetry.
Dipole Moment (μ)
- Cis-isomer: Usually has a higher dipole moment. Because the polar groups are on the same side, their individual bond dipoles add up.
- Trans-isomer: Usually has a zero or very low dipole moment. Since the groups are on opposite sides, the bond dipoles cancel each other out due to symmetry.
- Example: In 1,2-dichloroethene, the Cis form is polar (μ = 1.90D), while the Trans form is non-polar (μ = 0).
Boiling Point (B.P.)
- Cis > Trans: Generally, the Cis isomer has a higher boiling point.
- Reason: Higher polarity in Cis leads to stronger intermolecular dipole-dipole attractions. Stronger “molecular glue” means you need more heat to turn the liquid into gas.
Melting Point (M.P.)
- Trans > Cis: This is the opposite of the boiling point rule!
- Reason: It’s all about Symmetry and Packing. The Trans isomer is more symmetrical and linear, allowing the molecules to pack tightly into a crystal lattice—like neatly stacked bricks. Cis isomers are “U-shaped” and don’t fit together as well.
- Example: Maleic Acid (Cis): M.P. 131°C
- Fumaric Acid (Trans): M.P. 287°C (Significant difference!)
Solubility
- Cis > Trans: Generally, the Cis isomer is more soluble in polar solvents like water.
- Reason: Higher polarity and lower lattice energy (easier to break the crystal apart) make it easier for water molecules to surround and dissolve the Cis form.
Chemical Properties of Geometrical Isomers
While they share the same functional groups, their chemical reactivity can vary based on how close those groups are to each other.
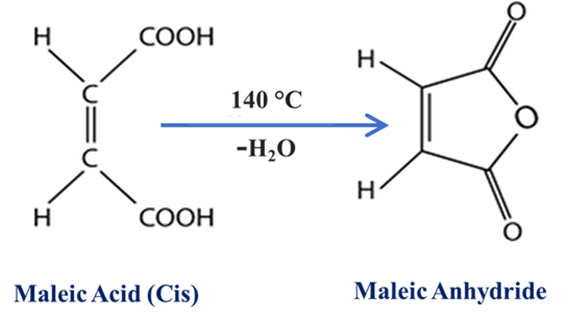
Formation of Cyclic Anhydrides
This is a classic “lab-bench” test to distinguish between isomers.
- Cis-isomers: When heated, the two carboxylic groups are close enough to react, lose a water molecule (H2O), and form a ring (Anhydride).
- Trans-isomers: They generally do not form anhydrides under the same conditions because the reactive groups are too far apart.
- Example: Maleic acid (Cis) easily forms Maleic anhydride at 160°C. Fumaric acid (Trans) requires much higher temperatures (≈300°C) because it must first isomerize to the Cis-form before it can react.
Stability (Steric Hindrance)
- Trans is more stable than Cis. In the Cis form, large groups are “crowded” on the same side, leading to Van der Waals strain (they literally bump into each other). In Trans, they are “socially distanced,” leading to lower energy and higher stability.
The Ultimate Comparison: Maleic Acid vs. Fumaric Acid:
These two molecules have the exact same molecular formula (C4H4O4) and the same functional groups (two carboxylic acids), but they are worlds apart.
Pharmaceutical Significance: Why Geometry Matters?
In Pharmacy, geometry is everything. The spatial arrangement affects how a drug “fits” into a biological receptor.
Diethylstilbestrol (DES)
This is perhaps the most famous example of GI in pharmacy. DES is a synthetic estrogen.
- The Trans-Isomer: This isomer is highly active. Its shape perfectly mimics the natural hormone estradiol, allowing it to bind to estrogen receptors.
- The Cis-Isomer: This isomer has only about 7% of the activity of the trans-form. Because the groups are on the same side, the molecule is “bulky” in the wrong places and cannot fit into the receptor “lock.”
Maleic Acid vs. Fumaric Acid
These two are simple but provide a great lesson in toxicity and metabolism.
- Fumaric Acid (Trans): This is a natural intermediate in the Krebs Cycle (Citric Acid Cycle). Our bodies recognize it and use it to produce energy.
- Maleic Acid (Cis): This isomer is toxic to the kidneys and is not a natural metabolite. Even though they have the same formula, the “Cis” geometry makes it behave entirely differently in a biological system.
Vitamin A (Retinal) and the Chemistry of Vision
Our ability to see depends entirely on a geometrical “flip.”
- 11-cis-retinal: In the dark, our eyes store Vitamin A in the Cis configuration, bound to a protein called opsin.
- All-trans-retinal: When light hits the retina, it triggers a photochemical reaction that converts the Cis isomer into the Trans isomer. This change in shape sends an electrical signal to the brain, allowing us to “see.”
Fatty Acids: Oleic vs. Elaidic Acid
This is relevant for students interested in Pharmaceutics and Formulation.
- Oleic Acid (Cis): Found in olive oil. The “Cis” double bond creates a “kink” in the chain, preventing the molecules from packing tightly. This is why it is a liquid at room temperature.
- Elaidic Acid (Trans): This “Trans” fat has a straight chain, allowing molecules to pack like bricks. It is a solid. This affects how these fats are absorbed and how they affect cholesterol levels.
Final Summary: Mastering the Flip
Shape is the Language of Pharmacy
We’ve journeyed through the rigid bonds of alkenes to discover that “Cis” and “Trans” are more than just prefixes—they are the blueprints for how a molecule melts in a lab, dissolves in a beaker, and reacts in a human cell. From the high melting point of Fumaric acid to the toxic nature of Maleic acid, geometry is the invisible hand guiding pharmacology.
Geometry is Destiny
The “Cis” or “Trans” label on a drug molecule isn’t just a naming convention—it’s a blueprint for its biological fate. Whether it’s the potency of a hormone or the way your eyes perceive light, the spatial arrangement of atoms dictates the success of a pharmaceutical therapy.
Bridging Chemistry and Dosage
For a pharmacist, knowing that a Trans isomer has a higher melting point might influence whether a drug is made into a heat-stable tablet or a liquid suspension.