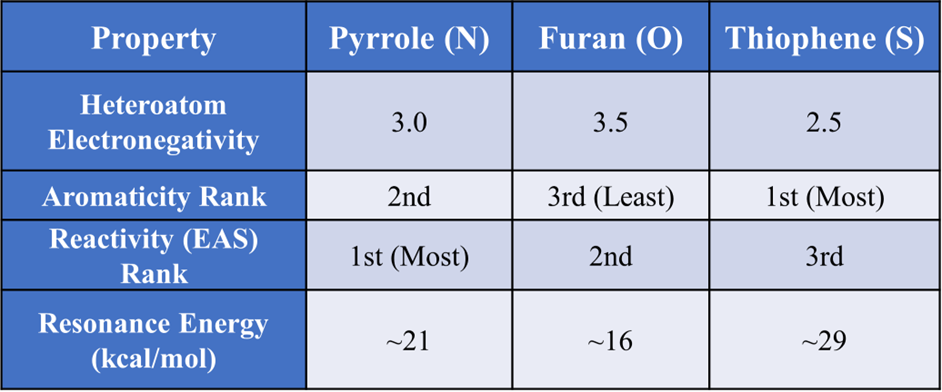
The relative aromaticity and reactivity of the five-membered heterocyclic compounds—Pyrrole, Furan, and Thiophene—are governed by the electronegativity of the heteroatom and the effectiveness of the p-orbital overlap with the carbon atoms.
Relative Aromaticity
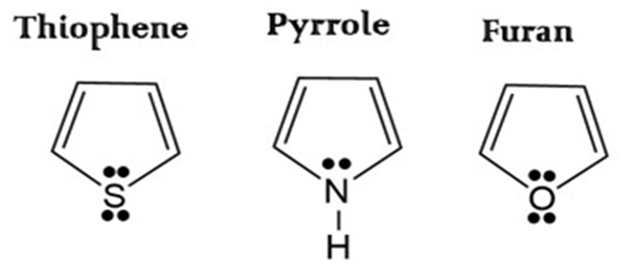
Aromaticity depends on how much a molecule “prefers” to keep its lone pair of the π -electrons delocalized into the ring to complete the (4n + 2) π-electron system (Huckel’s rule). They have 6 π electrons (4 from the two double bonds and 2 from a lone pair of the heteroatom) involved in their aromatic systems.
The aromaticity of these compounds is determined by the extent to which the lone pair of electrons on the heteroatom can delocalize into the ring to form a stable 6π-electron system. This delocalization is influenced by the electronegativity and orbital size of the heteroatom.
Order of Aromaticity: Thiophene (S) > Pyrrole (N) > Furan (O)
To memorize, it can be imagined as the snow (SNO).
- Thiophene: Thiophene is the most aromatic. Sulfur is in the third period of the periodic table. The lone pair of electrons on Sulphur is less tightly held and more readily available for delocalization into the ring. Thus Sulfur is less electronegative than oxygen and nitrogen. It “lets go” of its lone pair most easily to contribute to the (4n+2) π Huckel system. More importantly, the overlap between the 3p orbital of Sulfur and the 2p orbitals of Carbon is less efficient than 2p-2p overlap, but because Sulfur holds its electrons less tightly, the ring is more “benzene-like.” Thiophene has a higher resonance energy (approx. 29 kcal/mol) compared to pyrrole and furan, indicating greater stability and aromatic character.
- Pyrrole: Nitrogen is more electronegative than Sulfur but less than Oxygen. It shares its lone pair relatively well, making Pyrrole significantly aromatic, though less so than Thiophene. Pyrrole’s resonance energy is about 22 kcal/mol, placing it between thiophene and furan.
- Furan: Oxygen is the most electronegative. It holds its lone pair very tightly, resisting delocalization into the ring. Consequently, Furan has the least aromatic character and often behaves like a conjugated diene. The strong inductive electron-withdrawing effect of oxygen (-I effect) also competes with its mesomeric electron-donating effect (+M effect), reducing the overall electron density in the ring. Furan has the lowest resonance energy (approx. 16 kcal/mol) among the three, indicating it is the least aromatic and least stable.
Relative Reactivity (Electrophilic Substitution)
Since these rings are electron-rich due to the delocalization of the lone pair, they all undergo Electrophilic Aromatic Substitution (EAS) much faster than benzene.
Order of Reactivity: Pyrrole > Furan > Thiophene > Benzene
To memorize, it can be imagined as the nose (NOS).
- Pyrrole: The Nitrogen atom is the most efficient at “donating” its lone pair into the pi-system to stabilize the intermediate carbocation. This is referred as electron-donating resonance effect (+M effect). It significantly increases the electron density on the carbon atoms of the ring. The resonance structures show a delocalized negative charge on the ring carbons, making them highly susceptible to electrophilic attack. This makes Pyrrole exceptionally reactive—often compared to aniline or phenol. Electrophilic attack preferentially occurs at the C-2 (alpha) position because it leads to a more stable intermediate carbocation with more resonance structures (3 resonance structures) compared to attack at C-3 (2 resonance structures).
- Furan: Oxygen also has a +M effect. Its higher electronegativity causes it to also exert a significant -I (inductive electron-withdrawing) effect. The -I effect of oxygen is stronger than its +M effect in influencing the overall electron density on the ring carbons. Its make them less electron-rich than in pyrrole. Despite being the least aromatic, Furan is less activated towards electrophilic substitution reactions than pyrrole. It is more reactive than Thiophene because Oxygen is smaller and provides better orbital overlap for the transition state than Sulfur’s 3p orbitals. Furan is very susceptible to polymerization and ring-opening reactions under acidic conditions.
- Thiophene: Thiophene is the least reactive of the three towards electrophilic substitution, largely due to its higher aromaticity (greater resonance stability). The electronegativity of sulfur is closer to carbon, meaning its inductive effect is less pronounced than oxygen or nitrogen. Therefore it is less eager to react than the others. However, it is still much more reactive than benzene.
Summary Comparison Table
In conclusion, the interplay of electronegativity, orbital size and the balance between inductive and mesomeric effects of the heteroatom dictates the relative aromaticity and reactivity of these important heterocyclic compounds.